Abstract
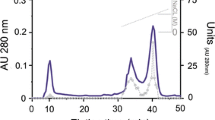
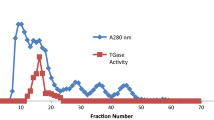
Isoelectric focusing revealed three enolase isoforms in pig brain, which were designated as αα- (pI = 6.5), αγ- (pI = 5.6), and γγ-enolase (pI = 5.2). The pI of purified γγ-enolase was also 5.2. The γγ-enolase isoform of enolase was purified from pig brain by a purification protocol involving heating to 55°C for 3 min, acetone precipitation, ammonium sulfate precipitation (40%–80%), DEAE Sephadex ion-exchange chromatography (pH 6.2), and Sephadex G200 gel filtration. The final specific activity was 82 units/mg protein. As with other vertebrate enolases, γγ-enolase from pig proved to be a dimer with a native mass of 85 kDa and a subunit mass of 45 kDa. The pH optimum for the reaction in the glycolytic direction is 7.2. The K m values for 2-PGA, PEP, and Mg2+ were determined to be 0.05, 0.25, and 0.50 mM, respectively, similar to K m values of other vertebrate enolases. The amino acid composition of pig γγ-enolase, as determined by amino acid analysis, shows strong similarity to the compositions of γγ-enolases from rat, human, and mouse, as determined from their amino acid sequences. Despite the differences seen with some residues, and considering the ways that the compositions were obtained, it is assumed that pig γγ-enolase is more similar than the composition data would indicate. Moreover, it is likely that the sequences of pig γγ-enolase and the other γγ-enolases are almost identical. Li+ proved to be a noncompetitive inhibitor with either 2-PGA or Mg2+ as the variable substrate. This enolase crystallized in the monoclinic space group P2, or P21. An R symm <5% was obtained for data between 50 and 3.65 Å, but was a disappointing 30% for data between 3.65 and 3.10 Å, indicating crystal disorder.
Similar content being viewed by others
REFERENCES
Aoki, T., Tanaka, T., and Watabe, H. (1992). Chem. Pharm. Bull. 40, 1236–1239.
Babbit, P. C., Hasson, M. S., Wedekind, J. E., Palmer, D. R., Barrett, W. C., Reed, G. H., Rayment, I., Ringe, D., Kenyon, G. L., and Gerlt, J. A. (1996). Biochemistry 35, 16489–16501.
Behnke, J. N., Massey, T. M., Dagher, S. M., and Deal, W. C., Jr. (1975). Analyt. Biochem. 69, 1–9.
Brown, C. K., Slates, K., Mattingly, S., Calie, P., and Farrar, W. (1997). Protein Sci. 6(Suppl. 1), 56.
Cali, L., Feo, S., Oliva, D., and Giallongo, A. (1990). Nucleic Acid Res. 18, 1893.
Duequerroy, S., Camus, C., and Janin, J. (1995). Biochemistry 34, 12513–12523.
Elliott, J. L., and Brewer, J. M. (1980). J. Inorg. Biochem. 12, 323–334.
Farrar, W. W., and Deal, W. C., Jr. (1995). J. Protein Chem. 14, 487–497.
Fletcher, L., Rider C. C., and Taylor, C. B. (1976). Biochim. Biophys. Acta 452, 245–252.
Giallongo, A., Oliva, D., Cali, L., Barba, G., Barbieri, G., and Feo, S. (1990). Eur. J. Biochem. 190, 567–573.
Ishioka, N., Isobe, T., Okuyama, T., and Nakajima, T. (1984). J. Biochem. 95, 611–617.
Kaghad, M., Dumont, X., Chalon, P., Lilias, J. M., Lamande, N., Lucas, M., Lazar, M., and Caput, D. (1990). Nucleic Acid Res. 18, 3638.
Kato, K., Shimizu, A., and Nagaya, M. (1987). Jpn. J. Cancer 78, 1100–1104.
Keller, A., Scama, H., Mermet, A., and Pujol, J.-F. (1981). J. Neurochem. 36, 1389–1397.
Kim, R. Y., and Wistow, G. J. (1993). FASEB J. 7, 464–469.
Kornblatt, M. J. (1996). Arch. Biochem. Biophys. 330, 12–18.
Kornblatt, M. J., and Klugerman, A. (1989). Biochem. Cell Biol. 67, 103–107.
Kornblatt, M. J., and Musil, R. (1990). Arch. Biochem. Biophys. 277, 301–305.
Lebioda, L., Stec, B., and Brewer, J. M. (1989). J. Biol. Chem. 264, 3685–3693.
Marangos, P. J., Zomzely-Neurath, C., Luk, D. C. M., and York, C. (1975). J. Biol. Chem. 250, 1884–1891.
Martin, R. G., and Ames, B. N. (1961). J. Biol. Chem. 236, 1372–1379.
McPherson, A. (1989). Preparation and Analysis of Protein Crystals, Krieger Malabar, Florida, pp. 83–159.
Nazarian, K. B., Simonian, S. Z., Kazarian, B. A., and Periz, P. (1995). Biokhimia 60, 746–753.
Oh, S.-K., and Brewer, J. M. (1973). Arch. Biochem. Biophys. 157, 491–499.
Peak, M. J., Peak, J. G., Stevens, F. J., Blamey, J., Mai, X., Zhou, Z. H., and Adams, M. W. W. (1994). Arch. Biochem. Biophys. 313, 280–286.
Py, B., Higgins, C. F., Krisch, H. M., and Carpousis, A. J. (1996). Nature 381, 169–172.
Ray, R., and Miller, D. M. (1991). Mol. Cell. Biol. 11, 2154–2161.
Ray, R. B., and Steele, R. (1997). Gene 186, 175–180.
Rider, C. C., and Taylor, C. B. (1974). Biochim. Biophys. Acta 365, 285–300.
Rider, C. C., and Taylor, C. B. (1975). Biochem. Biophys. Res. Commun. 66, 814–820.
Sakimura, K., Kushiya, E., Takahashi, Y., and Suzuki, Y. (1987). Gene 60, 103–113.
Schurig, H., Rutkat, K., Rachel, R., and Jaenicke, R. (1995). Protein Sci. 4, 228–236.
Scopes, R. K. (1977a). Biochem. J. 161, 253–263.
Scopes, R. K. (1977b). Biochem J. 161, 265–277.
Shimizu, A., Suzuki, F., and Kato, K. (1983). Biochem. Biophys. Acta 748, 278–284.
Singh, R. P., and Setlow, P. (1978). J. Bacteriol. 134, 353–355.
Suzuki, F., Umeda, Y., and Kato, K. (1980). J. Biochem. 87, 1587–1594.
Van Obberghen, E., Kamholz, J., Bishop, J. G., III, Zomzely-Neurath, C., and Lazzarini, R. A. (1988). J. Neurosci. Res. 19, 450–456.
Veronese, F. M., Schiavon, O., Boccu, E., Benassi, C. A., and Fontana, A. (1984). Int. J. Pept. Protein Res. 24, 557–562.
Viallard, J. L., Ven Murthy, M. R., and Dastugue, B. (1988). Neurochem. Sci. 13, 31–35.
Warburg, O., and Christian, W. (1941). Biochem. Z. 310, 384–421.
Weber, K., and Osborn, M. (1969). J. Biol. Chem. 244, 4406–4412.
Wedekind, J. E., Poyner, R. R., Reed, G. H., and Rayment, I. (1994). Biochemistry 33, 9333–9342.
Wold, F. (1971). Enolase in The Enzymes, 3rd ed., Vol. V (Boyer, P. D., ed.), Academic Press, New York, pp. 499–538.
Wold, F. (1975). Meth. Enzymol. 42C, 329–334.
Wolna, E., Wolny, M., and Baronowski, T. (1971). Acta Biochem. Polon. 18, 87–92.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Gorsich, S.W., Barrows, V., Halbert, J. et al. Purification and Properties of γγ-Enolase from Pig Brain. J Protein Chem 18, 103–115 (1999). https://doi.org/10.1023/A:1020659802760
Published:
Issue Date:
DOI: https://doi.org/10.1023/A:1020659802760