Abstract
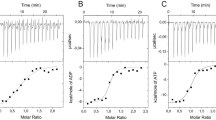
Selective treatment of pig kidney fructose 1,6-bisphosphatase with cyanate leads to the formation of an active carbamoylated derivative that shows no cooperative interaction between the AMP-binding sites, but completely retains the sensitivity to the inhibitor. By an exhaustive carbamoylation of the enzyme a derivative is formed that has a complete loss of cooperativity and a decrease of sensitivity to AMP. It was proposed that the observed changes of allosteric properties were due to the chemical modification of two lysine residues per enzyme subunit [Slebe et al. (1983), J. Protein Chem. 2, 437–443]. Studies of the temperature dependence of AMP sensitivity and the interaction with Cibacron Blue Sepharose of carbamoylated fructose 1,6-bisphosphatase derivatives indicate that the lysine residue involved in AMP sensitivity is located at the allosteric AMP site, while the lysine residue involved in AMP cooperativity is at a distinct location. Using [14C]cyanate, we identified both lysine residues in the primary structure of the enzyme; Lys50 is essential for AMP cooperativity and Lys112 appears to be the reactive residue involved in the AMP sensitivity. According to the fructose 1,6-bisphosphatase crystal structure, Lys50 is strategically positioned at the C1–C2 interface, near the molecular center of the tetramer, and Lys112 is in the AMP-binding site. The results reported here, combined with the structural data of the enzyme, strongly suggest that the C1ndash;C2 interface is critical for the propagation of the allosteric signal among the AMP sites on different subunits.
Similar content being viewed by others
REFERENCES
Benkovic, S. J., and de Maine, M. M. (1982). Adv. Enzymol. 53, 45–82.
Cárcamo, J. G., Yañez, A., Ludwig, H., Pinto, R., León, O., Reyes, A. M. and Slebe, J. C. (n.d.). Submitted for publication.
Chen, M., Chen, L., and Fromm, H. J. (1994). J. Biol. Chem. 269, 5554–5558.
Choe, J.-Y., Poland, B. W., Fromm, H. J., and Honzatko, R. B. (1998). Biochemistry 37, 11441–11450.
Colombo, G., and Marcus, F. (1974). Biochemistry 13, 3085–3091.
Colombo, G., Hubert, E., and Marcus, F. (1972). Biochemistry 11, 1798–1803.
Crestfield, A. M., Moore, S., and Stein, W. H. (1963). J. Biol. Chem. 238, 622–627.
Cruz, Z. M., Tanizaki, M. M., El-Dorry, H. A., and Bacila, M. (1979). Arch. Biochem. Biophys. 198, 424–433.
El-Maghrabi, M. R., Austin, L. R., Correia, J. J., and Pilkis, S. J. (1992). J. Biol. Chem. 267, 6526–6530.
Hubert, E., Ojeda, A., Reyes, A., and Slebe, J. C. (1986). Arch. Biochem. Biophys. 250, 336–344.
Ke, H., Zhang, Y., and Lipscomb, W. N. (1990a). Proc. Natl. Acad. Sci. USA 87, 5243–5247.
Ke, H., Thorpe, C. M., Seaton, B. A., Lipscomb, W. N., and Marcus, F. (1990b). J. Mol. Biol. 212, 513–539; 214, 950
Ke, H., Liang, J.-Y., Zhang, Y., and Lipscomb, W. N. (1991a). Biochemistry 30, 4412–4420.
Ke, H., Zhang, Y., Liang, J.-Y., and Lipscomb, W. N. (1991b). Proc. Natl. Acad. Sci. USA 88, 2989–2993.
Kurbanov, F., Choe, J.-Y., Honzatko, R. B., and Fromm, H. J. (1998). J. Biol. Chem. 273, 17511–17516.
Laemmli, U. K. (1970). Nature 227, 680–685.
Leyton, J. F., Chinelato, A. M., El-Dorry, H. A., and Bacila, M. (1980). Arch. Biochem. Biophys. 202, 168–171.
Liang, J.-Y., Huang, S., Zhang, Y., Ke, H., and Lipscomb, W. N. (1992). Proc. Natl. Acad. Sci. USA 89, 2404–2408.
Liang, J.-Y., Zhang, Y., Huang, S., and Lipscomb, W. N. (1993). Proc. Natl. Acad. Sci. USA 90, 2132–2136
Liu, F., and Fromm, H. J. (1990). J. Biol. Chem. 265, 7401–7406.
Lu, G., Stec, B., Giroux, E. L., and Kantrowitz, E. R. (1996). Protein Sci. 5, 2333–2342.
Lu, G., Giroux, E. L., and Kantrowitz, E. R. (1997). J. Biol. Chem. 272, 5076–5081.
Marcus, F. (1975). Biochemistry 14, 3916–3921.
Marcus, F. (1967). Arch. Biochem. Biophys. 122, 393–399.
Marcus, F. (1976). Biochemistry 15, 3505–3509.
Marcus, F., and Hubert, E. (1968). J. Biol. Chem. 243, 4923–4925.
Marcus, F., Edelstein, I., Reardon, I., and Heinrikson, R. L. (1982). Proc. Natl. Acad. Sci. USA 79, 7161–7165.
Nimmo, H. G., and Tipton, K. F. (1975). Eur. J. Biochem. 58, 567–574.
Pilkis, S. J., and Claus, T. H. (1991). Annu. Rev. Nutr. 11, 465–515.
Pontremoli, S., Grazi, E., and Accorsi, A. (1966). Biochemistry 5, 3568–3574.
Reyes, A., Burgos, M. E., Hubert, E., and Slebe, J. C. (1987). J. Biol. Chem. 262, 8451–8454.
Reyes, A., Rodríguez, P., and Slebe, J. C. (1992). Biochem. Int. 26, 347–356.
Reyes, A., Bravo, N. Ludwig, H., Iriarte, A., and Slebe, J. C. (1993). J. Protein Chem. 12, 159–168.
Riquelme, P., and Czarnecki, J. J. (1983). J. Biol. Chem. 258, 8240–8245.
Shyur, L.-F., Zhang, R., and Fromm, H. J. (1995). Arch. Biochem. Biophys. 319, 123–127.
Shyur, L.-F., Aleshin, A. E., Honzatko, R. B., and Fromm, H. J. (1996a). J. Biol. Chem. 271, 3005–3010.
Shyur, L.-F., Aleshin, A. E., Honzatko, R. B., and Fromm, H. J. (1996b). J. Biol. Chem. 271, 33301–33307.
Shyur, L.-F., Poland, B. W., Honzatko, R. B., and Fromm, H. J. (1997). J. Biol. Chem. 272, 26295–26299.
Slebe, J. C., Ojeda, A., Hubert, E., and Maccioni, R. (1981). In Molecular Approaches to Gene Expression and Protein Structure (Siddiqui, M. A. Q., Krauskopf, M., and Weissbach, H., eds.), Academic Press, New York, pp. 329–363.
Slebe, J. C., Herrera, R., Hubert, E., Ojeda, A., and Maccioni, R. (1983). J. Protein Chem. 2, 437–443
Stark, G. R. (1972). In Methods in Enzymology, Vol. 25B (Hirs, C. H. W., and Timasheff, S. N., eds.), Academic Press, New York, pp. 574–584.
Suda, H., Xu, G.-J., Kutny, R. M., Natalini, P., Pontremoli, S., and Horecker, B. L. (1982). Arch. Biochem. Biophys. 217, 10–14.
Taketa, K., and Pogell, B. M. (1965). J. Biol. Chem. 240, 651–662.
Tejwani, G. A. (1983). Adv. Enzymol. 54, 121–194.
Van Schaftingen, E. (1987). Adv. Enzymol. 59, 315–395.
Villeret, V., Huang, Sh., Fromm, H. J., and Lipscomb, W. N. (1995a). Proc. Natl. Acad. Sci. USA 92, 8916–8920.
Villeret, V., Huang, S., Zhang, Y., and Lipscomb, W. N. (1995b). Biochemistry 34, 4307–4315.
Xu, G. J., Datta, A. G., Singh, V. N., Suda, H., Pontremoli, S., and Horecker, B. L. (1981). Arch. Biochem. Biophys. 210, 98–103.
Xue, Y., Huang, S., Liang, J.-Y., Zhang, Y., and Lipscomb, W. N. (1994). Proc. Natl. Acad. Sci. USA 91, 12482–12486.
Zhang, Y., Liang, J.-Y., Huang, S., Ke, H., and Lipscomb, W. N. (1993). Biochemistry 32, 1844–1857.
Zhang, Y., Liang, J.-Y., Huang, S., and Lipscomb, W. N. (1994). J. Mol. Biol. 244, 609–624.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ludwig, H.C., Herrera, R., Reyes, A.M. et al. Suppression of Kinetic AMP Cooperativity of Fructose-1,6-Bisphosphatase by Carbamoylation of Lysine 50. J Protein Chem 18, 533–545 (1999). https://doi.org/10.1023/A:1020647116022
Published:
Issue Date:
DOI: https://doi.org/10.1023/A:1020647116022