Abstract
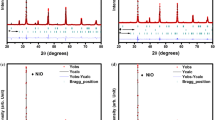
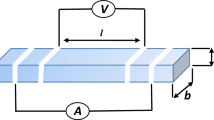
Y0.6Sr0.4Mn1−y Co y O3 (0 ≤ y ≤ 0.4) perovskite oxides were prepared by the coprecipitation method. The effect of Co substitution for Mn on the crystal structure, electrical conductivity and thermal expansion properties were investigated. By X-ray powder diffraction, the crystal structure was found to change from hexagonal symmetry of Y0.8Sr0.2MnO3 to orthorhombic of Y0.6Sr0.4Mn1−y Co y O3. The differences in the structure of the unsubstituted Y1−x Sr x MnO3 (0.2 ≤ x ≤ 0.4) are attributed to the average ionic radii of the cations and the amounts of Mn4+ present. The results of electrical conductivity analysis can be described by the small polaron hopping conductivity model. With Co substitution, the activation energy increases, possibly due to an increase of Jahn–Teller distortion, at an extent higher than the increase of the concentration of charge carriers; thus, the electrical conductivity decreases. In addition, the relative densities of the materials reached ∼94% with sintering at 1350°C for 12 h and had higher concentration of the available lattice sites, thus showing higher conductivity, than that with sintering at 1300°C for 6 h, which achieved ∼70% relative density. It is also found that the thermal expansion coefficient (TEC) increases as the Sr and Co content of Y1−x Sr x Mn1−y Co y O3 increases and those with Co content of y = 0.2 exhibit TEC compatibility with YSZ.
Similar content being viewed by others
References
P. J. GELLINGS and H. J. M. BOUWMEESTER, in “The CRC Handbook of Solid State Electrochemistry” (CRC Press, New York, 1997) p. 407.
J. H. KUO, H. U. ANDERSON and D. M. SPARLIN, J. Solid State Chem. 87 (1990) 55.
H. U. ANDERSON, Solid State Ionics 52 (1992) 33.
O. YAMAMOTO, Y. TAKEDA, R. KANNO and M. NODA, ibid. 22 (1987) 241.
B. FU and W. HUEBNER, Mater. Res. Soc. 9 (1994) 2645.
L. A. TIKHONOVA, P. P. ZHUK, A. A. VECHER and M. V. ZINKEVICH, Inorg. Mater. 28 (1992) 1535.
N. GUNASEKARAN, N. BAKSHI, C. B. ALCOCK and J. J. CARBERRY, Solid State Ionics 83 (1996) 145.
R. RAFFAELLE, H. U. ANDERSON, D. M. SPARLIN and P. E. PARRIS, Phys. Rev. Lett. 65 (1990) 1383.
J. W. STEVENSON, M. M. NASRALLAH, H. U. ANDERSON and D. M. SPARLIN, J. Solid State Chem. 102 (1993) 175.
J. A. ALONSO and M. J. MARTÍNEZ-LOPE, Inorg. Chem. 39 (2000) 917.
C. MOURE, M. VILLEGAS, J. F. FERNANDEZ, J. TARTAJ and P. DURAN, J. Mater. Sci. 34 (1999) 2565.
R. D. SHANNON, Acta Crtallogr. A 32 (1976) 751.
M. KERTESZ, I. RIESS, D. S. TANNHAUSER, R. LANGPAGE and F. J. ROHR, J. Solid State Chem. 42 (1982) 125.
M. H. HUANG, Ph. D Thesis, Imperial College, London (1991) p. 118.
J. B. GOODENOUGH, J. Appl. Phys. 37 (1966) 1415.
R. KOC and H. U. ANDERSON, J. Eur. Ceram. Soc. 15 (1995) 867.
E. POLLERT, S. KRUPICKA and E. KUZMICOVA, J. Phys. Chem. Solid 43 (1982) 1137.
N. GAYATHRI, A. K. RAYCHAUDHURI and S. K. TIWARY, Phys. Rev. B 56 (1997) 1345.
S. Y. BAE, D. J. SNYDER and S. X. WANG, J. Electr. Mater. 27 (1998) 1.
P. M. RACCAH and J. B. GOODENOUGH, Phys. Rev. 155 (1967) 932.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Huang, CY., Huang, TJ. Effect of Co substitution for Mn on Y1−x Sr x MnO3 properties for SOFC cathode material. Journal of Materials Science 37, 4581–4587 (2002). https://doi.org/10.1023/A:1020640114502
Issue Date:
DOI: https://doi.org/10.1023/A:1020640114502