Abstract
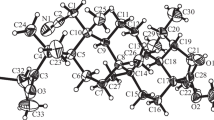
Addition of N-(2-mercaptopropyonyl)glycine to (+)-limonene or (-)-β-pinene in the presence of a Lewis acid involves the mercapto group; simultaneously, the carboxyl function is esterified by ethanol used as solvent. Both reactions give rise to a terpenoid of the menthane structure, which contains, along with a sulfide group, a peptide fragment promising in terms of biologic activity, as well as an ester group.
Similar content being viewed by others
REFERENCES
Wattenberg, L.W., Sparnins, V.L., and Barany, G., Cancer Res. 1989, vol. 49, no. 10, p. 2689.
Schaffer, E.M., Liu, J.-J., Green, J., Dangler, C.A., and Milner, J.A., Cancer Lett. 1996, vol. 102, p. 199.
Morgunova, V.A., Nikitina, L.E., Plemenkov, V.V., Chugunov, Yu.V., and Fazlyeva, M.G., Zh. Org. Khim. 2000, vol. 36, no. 4, pp. 512-514.
Nikitina, L.E., Plemenkov, V.V., Shaikhutdinov, R.A., and Klochkov, V.V., Zh. Org. Khim. 1996, vol. 32, no. 7, p. 1007.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Nikitina, L.E., Startseva, V.A., Vakulenko, I.A. et al. Synthesis of Polyfunctional Terpenoids from Monoterpenes and N-(2-Mercaptopropyonyl)glycine. Russian Journal of General Chemistry 72, 974–975 (2002). https://doi.org/10.1023/A:1020459013118
Issue Date:
DOI: https://doi.org/10.1023/A:1020459013118