Abstract
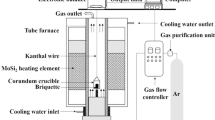
Studies on the mechanism and kinetics of the reaction leading to FeVMoO7 have been made using XRD and IR spectroscopy. The activation energy, E = 265 ± 13 kJ/mol has been calculated by the Erofeev model.
Similar content being viewed by others
REFERENCES
J. Walczak, M. Kurzawa, E. Filipek: J. Therm. Anal., 31, 271 (1986).
J. Walczak, J. Ziolkowski, M. Kurzawa, L. Trzesniowska: J. Therm. Anal., 29, 983 (1984).
J. Walczak, J. Ziolkowski, M. Kurzawa, L. Trzesniowska: Pol. J. Chem., 59, 713 (1985).
J. Walczak, M. Kurzawa, L. Trzesniowska: Thermochim. Acta, 92, 567 (1985).
J. Walczak, M. Kurzawa: Thermochim. Acta, 127, 363 (1988).
J. Walczak, M. Kurzawa: J. Therm. Anal., 31, 513 (1986).
J. Walczak, M. Kurzawa, P. Tabero: Thermochim. Acta, 118, 1 (1987).
J. Walczak, M. Kurzawa, E. Filipek: J. Therm. Anal., 32, 1751 (1987).
J. Walczak, M. Kurzawa, E. Filipek: Thermochim. Acta, 117, 9 (1987).
M. Kurzawa: Thermochim. Acta, 189, 129 (1991).
J. Walczak, M. Kurzawa, P. Tabero: J. Therm. Anal., 33, 969 (1988).
X. Wang, K.R. Heier, C.L. Stern, K.R. Poeppelmeier: Inorg. Chem., 37, 3252 (1998).
J. Beretka, T. Brown: J. Am. Cer. Soc., 66, 383 (1983).
J. Deren, J. Haber, R. Pampuch: Chemia Ciala Stalego, p.629, PWN, Warszawa 1975.
W. Yan, C. Ma, J. Wu, W. Zhang, D. Jang: J. Therm. Anal. Cal., 58, 393 (1999).
K. Eda: J. Solid State Chem., 95, 64 (1991).
M. Kurzawa, J. Mater. Sci. Lett., 11, 976 (1992).
P. Forzatti, C.M. Mari, P-L. Villa: Mat. Res. Bull., 22, 1593 (1987).
L. Stanescu, I. Ardelean, E. Burzo: Solid State Commun., 32, 1289 (1979).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Tabero, P., Filipek, E. Studies on the kinetics and mechanism of the synthesis of FeVMoO7 in the solid state. Reaction Kinetics and Catalysis Letters 77, 21–27 (2002). https://doi.org/10.1023/A:1020327416499
Issue Date:
DOI: https://doi.org/10.1023/A:1020327416499