Abstract
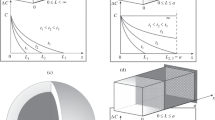
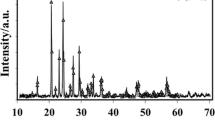
A mathematical diffusion model, which takes into account the electrochemical behavior of partially-discharged thin-layer electrodes made of intercalation materials upon interruption of circuit, is put forward. The applicability of the model is tested by the example of Li x V2O5 films. According to theoretical calculations and experimental data, the equilibrium potential of the films studied depends practically linearly on the degree of intercalation with a slope of –0.8 V for intercalation degrees of 0.3–0.7. The chemical diffusion coefficient of lithium in the films is equal to 1.5 × 10–11 cm2/s and changes insignificantly at these intercalation degrees.
Similar content being viewed by others
REFERENCES
Su-Il Pyun and Loon-Sang Bac, Electrochim. Acta, 1996, vol. 41, p. 919.
John Wen, C., Boucamp, B.A., and Huggins, R.A., J. Electrochem. Soc., 1997, vol. 126, p. 2258.
Sato, Y., Asaca, T., Tokugawa, H., and Kobayakawa, K., J. Power Sources, 1997, vol. 68, p. 674.
Pereira-Ramos, J.P. and Messina, R.M., Electrochim. Acta, 1998, vol. 33, p. 1003.
Lykov, A.V., Teoriya teploprovodnosti (A Theory of Heat Transfer), Moscow: Vysshaya Shkola, 1967.
Shembel, E., Apostolova, R., Nagirny, V., et al., J. Power Sources, 1999, vol. 81/82, p. 480.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Tysyachnyi, V.P., Shembel', E.M. & Apostolova, P.D. Electrochemical Behavior of Partially-Discharged Li x V2O5 Electrodes upon Interruption of Current. Russian Journal of Electrochemistry 38, 1004–1008 (2002). https://doi.org/10.1023/A:1020297129588
Issue Date:
DOI: https://doi.org/10.1023/A:1020297129588