Abstract
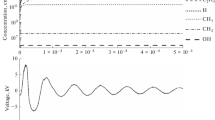
The kinetics of cyclohexane oxidation in a barrier discharge reactor was simulated for a single voltage pulse. A significant difference between the yields of O3 obtained experimentally (not detected) and theoretically (15.5 wt %) suggests that O3 was absent from the reaction mixture because of a fast reaction between O(3 P) and an excited cyclohexane molecule. This hypothesis was indirectly supported by experimental data on the oxidation of a mixture of n-hexane and cyclohexene (50 : 50 wt %). The integrated rate constant of the reaction of O(3 P) with n-hexane was 1.4 × 10–12 cm3/s, which is an order of magnitude higher than the published value 1.2 × 10–13 cm3/s.
Similar content being viewed by others
REFERENCES
Coogan, J.J. and Avtar, S.J., Silent Discharge Plasma (SDP) for Point-of-Use (POU) Abatement of Volatile Organic Compound (VOC) Emissions: Final Report (ESHC003), SEMATEC, 1998.
McCulla, W.C., Rososha, L.A., Neely, W.C., Clothi-aux, E.J., Kushner, M.J., and Rood, M.J., Proc. First INEL Plasma Applications to Waste Treatment Work-shop, Idaho Falls, 1991, p. 1.
Zlatopol'skii, V.M. and Smolenskaya, T.S., Khim. Fiz., 1997, vol. 16, no. 8, p. 105.
Bugaev, S.P., Kozyrev, A.V., Kuvshinov, V.A., Soch-ugov, N.S., and Khryapov, P.A., Dokl. Akad. Nauk, 1997, vol. 354, no. 2, p. 200.
Kozlov, K.V., Michel, P., and Wagner, H.-E., Contributed Papers, Int. Symposium on High Pressure Low Tempera-ture Plasma Chemistry HAKONE VII, Greifswald, 2000, vol. 2, p. 262.
Kudryashov, S.V., Shchegoleva, G.S., Sirotkina, E.E., and Ryabov, A.Yu., Khim. Vys. Energ., 2000, vol. 34, no. 2, p. 145 [High Energy Chem. (Engl. transl.), 2000, vol. 34, no. 2, p. 112].
Kudryashov, S.V., Ryabov, A.Yu., Sirotkina, E.E., and Shchegoleva, G.S., Khim. Vys. Energ., 2001, vol. 35, no. 2, p. 149 [High Energy Chem. (Engl. transl.), 2001, vol. 35, no. 2, p. 120].
Eliasson, B., Egli, W., and Kogelschatz, U., Pure Appl. Chem., 1994, vol. 66, no. 6, p. 1275.
Samoilovich, V.G., Gibalov, V.I., and Kozlov, K.V., Fiz-icheskaya khimiya bar'ernogo razryada (Physical Chemistry of a Barrier Discharge), Moscow: Mosk. Gos. Univ., 1989.
Polak, L.S., Neravnovesnaya khimicheskaya kinetika i ee primenenie (Nonequilibrium Chemical Kinetics and Its Applications), Moscow: Nauka, 1979.
Morgan, W.L., Kinema Software BOLSIG, Version 1.05, 1994.
Slovetskii, D.I., Khim. Plazmy, 1981, vol. 8, p. 189.
Chemical Kinetics Simulator v1.01, IBM, 1996.
DeMore, W.B., Sander, S.P., Golden, D.M., Hamp-son, R.F., Kurylo, M.J., Howard, C.J., Ravishan-kara, A.R., Kolb, C.E., and Molina, M.J., Chemical Kinetics and Photochemical Data for Use in Strato-spheric Modeling, JPL Publication 24-94, NASA, Jet Propulsion Laboratory, California Inst. of Technology, 1994, Eval. no 11.
Herron, J.T. and Huie, R.E., J. Phys. Chem. Ref. Data, 1973, vol. 2, no. 3, p. 467.
Paraskevopoulos, G. and Cvetanovic, R.J., J. Am. Chem. Soc., 1969, vol. 91, no. 27, p. 7572.
Kondrat'ev, V.N., Konstanty skorosti gazofaznykh reaktsii (Rate Constants of Gas-Phase Reactions), Mos-cow: Nauka, 1970.
Berezin, I.V., Denisov, E.T., and Emanuel', N.M., Okislenie tsiklogeksana (Oxidation of Cyclohexane), Moscow: Mosk. Gos. Univ., 1962.
Denisov, E.T., Konstanty skorosti gomoliticheskikh zhid-kofaznykh reaktsii (Rate Constants of Homolytic Liquid-Phase Reactions), Moscow: Nauka, 1971.
Razumovskii, S.D. and Zaikov, G.E., Ozon i ego reaktsii s organicheskimi soedineniyami (Ozone and Its Reac-tions with Organic Compounds), Moscow: Nauka, 1974.
Rusanov, V.D., Fridman, G.V., and Sholin, V.G., Usp. Fiz. Nauk, 1981, vol. 134, no. 2, p. 185.
Mayer, S.W. and Schieler, L., J. Phys. Chem., 1968, vol. 72, no. 7, p. 2628.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kudryashov, S.V., Shchegoleva, G.S., Ryabov, A.Y. et al. Simulation of the Kinetics of Cyclohexane Oxidation in a Barrier Discharge Reactor. High Energy Chemistry 36, 349–354 (2002). https://doi.org/10.1023/A:1020254818916
Issue Date:
DOI: https://doi.org/10.1023/A:1020254818916