Abstract
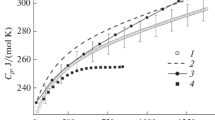
Measurements of the heat capacity by quasi-adiabatic, intermittent energy increments from 5 to 350 K show small high heat-capacity anomalies near 7 and 10 K which are attributed to superconducting transitions seen by magnetic measurements on the same carefully synthesized and well-characterized sample of (Hf0.934Zr0.057)(N0.97). Although no previous heat capacity measurements over the cryogenic region are known, the estimated 298.15 K standard entropy values (S/R) vary in the literature from about 200 per cent higher to 5 per cent lower than our measured value of (5.28±0.01)R−1 when the formula is represented as above. A simple scheme to represent and predict values based on both molar volumes and atomic masses for related materials is presented which seems more reliable on a limited sample than do others despite the intrusion of lanthanide contraction.
Similar content being viewed by others
References
B. Karlsson, R. P. Shimshock, B. O. Seraphin and J. C. Haygarth, Physica Scripta, 25 (1982) 775.
W. M. Latimer, J. Amer. Chem. Soc., 43 (1921) 818.
K. K. Kelley and E. G. King, The Metallurgy of Hafnium, D. T. Thomas and E. T. Hayes; Eds U. S. Atomic Energy Commission 1961, p. 323.
P. J. Spencer, Chapter in Hafnium: Physico-Chemical Properties of its Compounds and Alloys, Thermochemical Properties, K. L. Komarek; Ed., Atomic Energy Rev. Spec. Issue No. 8, IAEA, Vienna 1981, p. 5.
L. B. Pankratz, J. M. Stuve and N. A. Gokcen, Thermodynamic Data for Mineral Technology, USBM Bulletin 677, U.S. Department of the Interior 1984, p. 254.
I. Barin, Thermochemical Data of Pure Substances, VCH; Weinheim 1995, p. 817.
G. Grimvall, High Temperatures-High Pressures, 17 (1985) 607.
E. Rudy and F. Benesovsky, Montash. Chem., 92 (1961) 415.
L. E. Toth, Transition Metal Carbides and Nitrides, Vol. 9 of Refractory Material Sciences.
W. Lengauer and P. Ettmayer, Proceedings 12th International Plansee Seminar’ 89, Vol. 2, p. 497.
E. F. Westrum, Jr., G. T. Furukawa and J. P. McCullough, Experimental Thermodynamics, Vol. 1, J. P. McCullough and D. W. Scott; Eds, Butterworths, London 1968, p. 133.
E. F. Westrum, Jr., Proceedings NATO Advanced Study Institute on Thermochemistry, A. V. Ribeiro da Silva; Ed., Reidel, New York 1984, p. 745.
J. K. Hulm and B. T. Matthias, Chapter in Superconductor Science, S. Foners and B.B. Schwartz; editors, Plenum Press, New York 1981, p. 34.
T. Willis, Department of Materials Science, University of Wisconsin (Madison), Personal communication (January 1994).
N. Komada and E. F. Westrum, Jr., J. Chem. Thermodyn., 29 (1997) 311.
K. C. Mills, cited by O. Kubaschewski, C. B. Alcock and P. J. Spencer, Materials Thermochemistry, 6th Ed., Pergamon Press, Oxford 1993, p. 172.
Powder Refraction File, Inorganic Phases, © JCPDS International Centre for Powder Diffraction Data, Swarthmore, PA 19081 1981.
R. Hultgren, P. Desai, D. T. Hawkins, M. Gleiser, K. K. Kelley and D. D. Wagman, Selected Values of the Thermodynamic Properties of the Elements, American Society for Metals, Metals Park, OH 1973.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Westrum, E.F., Sommers, J.A. Heat capacity of hafnium mononitride from temperatures of 5 to 350 K: An estimation procedure. Journal of Thermal Analysis and Calorimetry 69, 103–112 (2002). https://doi.org/10.1023/A:1019937723458
Issue Date:
DOI: https://doi.org/10.1023/A:1019937723458