Abstract
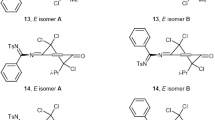
Halogenation of 4-aroyl(arylsulfonyl)oxyimino-2-methyl-2,5-cyclohexadienones yields Z,E-isomeric 4-aroyl(arylsulfonyl)oxyimino-5,6-dihalo-2-methyl-2-cyclohexenones and (E)-4-aroyl(arylsulfonyl)oxyimino-5,6-dihalo-6-methyl-2-cyclohexenones. Further chlorination leads to formation of (Z,E)-4-aroyl(arylsulfonyl)oxyimino-2,5,6-trichloro-6-methyl-2-cyclohexenones.
Similar content being viewed by others
REFERENCES
Avdeenko, A.P., Glinyanaya, N.M., Konovalova, S.A., Shishkina, S.V., and Goncharova, S.A., Russ. J. Org. Chem., 2002, vol. 38, no. 5, pp. 683-691.
Avdeenko, A.P., Glinyanaya, N.M., and Pirozhenko, V.V., Russ. J. Org. Chem., 1995, vol. 31, no. 10, pp. 1380-1385.
Avdeenko, A.P., Glinyanaya, N.M., and Pirozhenko, V.V., Russ. J. Org. Chem., 1996, vol. 32, no. 1, pp. 85-89.
Baldwin, J.E. and Norris, R.K., J. Org. Chem., 1981, vol. 46, no. 4, pp. 697-703.
Avdeenko, A.P., Zhukova, S.A., and Konovalova, S.A., Russ. J. Org. Chem., 2001, vol. 37, no. 3, pp. 382-387.
Avdeenko, A.P. and Glinyanaya, N.M., Russ. J. Org. Chem., 1995, vol. 31, no. 11, pp. 1507-1513.
Avdeenko, A.P., Velichko, N.V., and Romanenko, E.A., Zh. Org. Khim., 1991, vol. 27, no. 11, pp. 2350-2361.
Avdeenko, A.P., Glinyanaya, N.M., and Pirozhenko, Zh. Org. Khim., 1993, vol. 29, no. 7, pp. 1402-1411.
Avdeenko, A.P., Zhukova, S.A., Glinyanaya, N.M., and Konovalova, S.A., Russ. J. Org. Chem., 1999, vol. 35, no. 4, pp. 560-571.
Sheldric, G.M., SHELXS-86. Program for the Solution of Crystal Structures, Gottingen: Univ. of Gottingen, 1986.
Sheldric, G.M., SHELXL-93. Program for the Refinement of Crystal Structures, Gottingen: Univ. of Gottingen, 1993.
Titov, E.A. and Burmistrov, S.I., Ukr. Khim. Zh., 1960, vol. 26, no. 6, pp. 744-749.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Avdeenko, A.P., Glinyanaya, N.M., Konovalova, S.A. et al. Halogenation of N-Substituted p-Quinonimines and p-Quinone Oxime Esters: II. Chlorination and Bromination of 4-Aroyl(arylsulfonyl)oxyimino-2-methyl-2,5-cyclohexadienones. Russian Journal of Organic Chemistry 38, 692–698 (2002). https://doi.org/10.1023/A:1019663122800
Issue Date:
DOI: https://doi.org/10.1023/A:1019663122800