Abstract
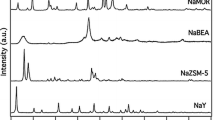
The influence of the temperature on the protonation of acetonitrile by acidic zeolites was studied by infrared spectroscopy. Acidity at room or low temperature was not correlated with the protonation temperature, but the zeolitic structure played an important role. A new technique is presented for the study of the acidity of solids under reaction conditions. A good correlation was obtained in a series of various zeolites between the catalytic activity in the cracking of n-hexane and the protonation temperature of acetonitrile.
Similar content being viewed by others
References
(a) H. Kn¨ozinger, in: Elementary Reaction Steps in Heterogeneous Catalysis, eds. R.W. Joynerand and R.A. van Santen (Kluwer, Dordrecht, 1993) p. 267; (b) J.A. Lercher, C. Gr¨undling and G. Eder-Mirth, Catal. Today 27 (1996) 353.
S. Jolly, J. Saussey and J.C. Lavalley, J. Mol. Catal. 86 (1994) 401.
M. Maache, A. Janin, J.C. Lavalley and E. Benazzi, Zeolites 15 (1995) 507.
L. Kubelkova, J. Kotrla and J. Florian, J. Phys. Chem. 99 (1995) 10285.
G.A. Olah and T.E. Kiovsky, J. Am. Chem. Soc. 90 (1968) 4666.
A. Corma, C.M. Zicovich-Wilson and P. Viruela,J. Phys. Chem. 98 (1994) 10863.
J.F. Haw, M.B. Hall, A.E. Alvarado-Swaisgood, E.J. Munson, Z. Lin, L.W. Beck and T. Howard, J. Am. Chem. Soc. 116 (1994) 7308.
C. Bourdillon, C. Geguen and M. Guisnet, Appl. Catal. 61 (1990) 123.
Y. Sun, P.J. Chu and J.H. Lunsford, Langmuir 7 (1991) 3027.
S. Jolly, J. Saussey, A. Janin, F. Thibault-Starzyk and J.C. Lavalley, J. Chim. Phys., in press.
F.H. Wielers, M. Waarkamp and M.F.M. Post, J. Catal. 127 (1991) 51.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Thibault-Starzyk, F., Travert, A., Saussey, J. et al. Correlation between activity and acidity on zeolites: a high temperature infrared study of adsorbed acetonitrile. Topics in Catalysis 6, 111–118 (1998). https://doi.org/10.1023/A:1019182826692
Issue Date:
DOI: https://doi.org/10.1023/A:1019182826692