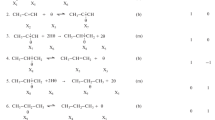Abstract
The hydrogenation of acetylene and acetylene–ethylene mixtures on a commercial palladium catalyst for hydrogenation of acetylene traces in ethylene‐rich feedstock was studied. The obtained results suggest that during the reaction two types of catalytic sites are created by carbonaceous deposits on the palladium surface: A sites representing small spaces and E sites representing large spaces of palladium between the carbonaceous deposits. The A sites adsorb acetylene and hydrogen but, due to steric hindrance, do not adsorb ethylene. Consequently, these sites are involved in the conversion of acetylene, producing ethylene and small amounts of ethane and butadiene, and they are inactive in the hydrogenation of ethylene. Instead, the E sites adsorb all reactants and, therefore, they are responsible for the hydrogenation of ethylene and butadiene at low acetylene partial pressures.
Similar content being viewed by others
References
A.N.R. Bos and K.R. Westerterp, Chem. Eng. Proc. 32 (1993) 1.
A. Borodziński, R. Duś, R. Frak, A. Janko and W. Palczewska, in: Proc. 8th Int. Congress on Catalysis, eds. P.B. Wells and G.C. Bond (The Chemical Society, London, 1977) paper A7.
J. Stachurski and A. Frackiewicz, J. Less Common Met. 108 (1985) 249.
S.B. Ziemecki, G.A. Jones, D.G. Swartzfager, R.L. Harlow and J. Faber, Jr., J. Am. Chem. Soc. 107 (1985) 4547.
J. Zieliński and A. Borodziński, Appl. Catal. 13 (1985) 305.
A. Borodziński, Polish J. Chem. 72 (1998) 2455.
S.J. Thomson and G. Webb, J. Chem. Soc. Chem. Commun. (1976) 526.
A.S. Al-Ammar and G. Webb, J. Chem. Soc. Faraday Trans. I 74 (1978) 195, 657; 75 (1979) 1900.
G. Webb, Catal. Today 7 (1990) 139.
J. Margitfalvi, L. Guczi and A.H. Weiss, J. Catal. 72 (1981) 185.
N.R.M. Sassen, A.J. den Hartog, F. Jongerius, J.F.M. Aarts and V. Ponec, Faraday Discuss. Chem. Soc. 87 (1989) 311.
A.J. den Hartog, M. Deng, F. Jongerius and V. Ponec, J. Mol. Catal. 60 (1990) 99.
J. Houzvicka, R. Pestman and V. Ponec, Catal. Lett. 30 (1995) 289.
A. Sárkány, L. Guczi and A.H. Weiss, Appl. Catal. 10 (1984) 369.
A. Sárkány, P. Sandor and L. Guczi, Appl. Catal. 12 (1984) 373.
S. LeViness, V. Nair, A.H. Weiss, Z. Schay and L. Guczi, J. Mol. Catal. 25 (1984) 131.
A. Borodziński, Polish J. Chem. 69 (1995) 111.
S. Asplund, J. Catal. 158 (1996) 267.
S. Asplund, C. Fornell, A. Holmgren and S. Inradoust, Catal. Today 24 (1995) 181.
M. Larsson, J. Jannson and S. Asplund, J. Catal. 178 (1998) 49.
I. Yasumori, T. Kabe and Y. Inoue, J. Phys. Chem. 78 (1974) 583.
A. Borodziński and A. Gołebiowski, in: Proc. 2nd Int. Symp. ISSHAC, Poland-Slovakia, 1995, p. 236.
A. Borodziński and A. Gołebiowski, Langmuir 13 (1997) 883.
D. Duca, F. Frusteri, A. Parmaliana and G. Deganello, Appl. Catal. A 146 (1996) 269.
D. Duca, D. Arena, A. Parmaliana and G. Deganello, Appl. Catal. A 172 (1998) 207.
M. Szukiewicz, A. Borodziński and A. Gołebiowski, Przem. Chem. 77 (1998) 340.
R. Duś, Surf. Sci. 50 (1975) 241.
R. Duś and W. Lisowski, Surf. Sci. 85 (1979) 183.
F. Zaera and G.A. Somorjai, J. Am. Chem. Soc. 106 (1984) 2288.
A. Sárkány, Z. Schay, G. Stefler, L. Borkó, J.W. Hightower and L. Guczi, Appl. Catal. A 124 (1995) L181.
A. Borodziński, A. Gołebiowski and B. Górecka, in: Book of Abstracts EuropaCat II Congr. (1997) p. 98.
J. Zieliński, React. Kinet. Catal. Lett. 17 (1978) 173.
J. Zieliński, J. Catal. 76 (1982) 157.
D.J. Suh and S.-K. Ihm, J. Catal. 149 (1994) 486.
N.E. Buyanova, N.B. Ibragimova, A.P. Karnaukhov and N.G. Koreleva, Kinet. Katal. 13 (1972) 295.
J.J.F. Scholten, A.P. Pijpers and A.M.L. Hustings, Catal. Rev. Sci. Eng. 27 (1985) 173.
A. Borodziński and M. Bonarowska, Langmuir 13 (1997) 5613.
A. Borodziński, Polish J. Chem. 68 (1994) 583.
F. Kapteijn and J.A. Moulijn, in: Handbook of Heterogeneous Catalysis, Vol. 3, eds. G. Ertl, H. Knözinger and J. Weitkamp (VCH, Weinheim, 1997) section 9.1.
A.N.R. Bos, E.S. Bootsma, F. Foeth, H.W.J. Sleyster and K.R. Westerterp, Chem. Eng. Proc. 32 (1993) 53.
S. Asplund, Ph.D. thesis, Chalmers University of Technology, Göteborg (1996); J. Catal. 158 (1996) 267.
C. Godinez, A.L. Cabanes and G. Villora, Canad. J. Chem. Eng. Proc. 74 (1996) 84.
V.A. Men'shikov, Yu.G. Fal'kovich and M.E. Aerov, Kinet. Catal. 16 (1975) 1335.
L.Z. Gva and K.E. Kho, Kinet. Catal. 29 (1988) 381.
M. Molero, B.F. Bartlett and W.T. Tysoe, J. Catal. 181 (1999) 49.
A. Sandell, A. Beutler, A. Jaworski, M. Wiklund, K. Heister, R. Nyholm and J.N. Andersen, Surf. Sci. 415 (1998) 411.
L.L. Kesmodel, G.D. Waddill and J.A. Gates, Surf. Sci. 138 (1984) 464.
W.T. Tysoe, G.L. Nyberg and R.M. Lambert, Surf. Sci. 135 (1983) 128
W.T. Tysoe, Langmuir 12 (1996) 78.
W.T. Tysoe, G.L. Nyberg and R.M. Lambert, J. Phys. Chem. 90 (1986) 3188.
G.A. Somorjai and F. Zaera, J. Phys. Chem. 86 (1982) 3070.
W.T. Tysoe, G.L. Nyberg and R.M. Lambert, J. Phys. Chem. 88 (1984) 1960.
P. Cremer, X. Su, Y.R. Shen and G.A. Somorjai, J. Am. Chem. Soc. 118 (1996) 2942.
M. Krawczyk, Ph.D. thesis, Dept. of Catal. on Metals, Inst. of Phys. Chem., Pol. Acad. Sci., Warsaw (1992).
W. Palczewska, I. Ratajczykowa, I. Szymerska and M. Krawczyk, in: Proc. 8th Int. Congress on Catalysis, Vol. 4 (Verlag-Chemie, Weinheim, 1984) p. 173.
M. Krawczyk, J. Sobczak and W. Palczewska, Catal. Lett. 17 (1993) 21.
L. Pauling, The Chemical Bond (Cornell University Press, Ithaca NY, 1997) pp. 88, 89, 136, 140, 152.
A. Borodziński, unpublished results.
A. Borodziński, Appl. Catal. A, submitted.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Borodziński, A. Hydrogenation of acetylene–ethylene mixtures on a commercial palladium catalyst. Catalysis Letters 63, 35–42 (1999). https://doi.org/10.1023/A:1019052618049
Issue Date:
DOI: https://doi.org/10.1023/A:1019052618049