Abstract
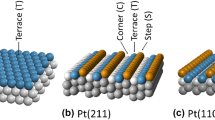
Some years ago, LEED studies of Ogletree et al. and Wander et al. found that benzene undergoes a structural rearrangement when the benzene adsorbs on platinum. In contrast, Horsley et al. proposed that the benzene adsorbs without undergoing significant distortion. In this paper UV/HREELS spectroscopy is used to examine benzene adsorption on Pt(110). We find that the benzene can adsorb into two different states: a weakly bound form of benzene with UV/HREELS spectrum similar to gas-phase benzene, and a strongly bound form of benzene with UV/HREELS and IR/HREELS spectra which are consistent with formation of a diene (e.g., 1,4-cyclohexadiene). A comparison of our data to the results of Wander et al. and Horsley et al. shows that the weakly bound form of benzene is consistent with Horsley’s assignments, while the strongly bound species looks as expected from the work of Wander et al. The conclusion from our study then is that Wander et al. and Horsley et al. are both correct. They are just studying different forms of benzene on the platinum surface.
Similar content being viewed by others
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Chen, N.C., Ford, L.P. & Masel, R.I. UV/HREELS spectroscopy of benzene on Pt(110). Catalysis Letters 56, 105–109 (1998). https://doi.org/10.1023/A:1019048810528
Issue Date:
DOI: https://doi.org/10.1023/A:1019048810528