Abstract
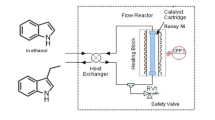
The one‐step procedure of amination of bifunctional secondary alcohols to diamines has been investigated in a continuous fixed‐bed reactor. Application of supercritical NH3 as a solvent and reactant suppressed catalyst deactivation and improved selectivities to amino alcohol intermediates, whereas selectivities to diamines remained poor (8–10%). The main reason for the low diamine selectivity of 1,3‐dihydroxy compounds is water elimination leading to undesired monofunctional products via α,β‐unsaturated alcohol, ketone or amine intermediates. This side reaction does not occur with 1,4‐dihydroxy compounds which afford high aminol and diamine selectivities under similar conditions. Amination of secondary diols with ammonia was found to be faster, but less selective than that of the corresponding primary 1,3‐propanediol.
Similar content being viewed by others
References
A. Fischer, T. Mallat and A. Baiker, Catal. Today 37 (1997) 167.
T. Mallat and A. Baiker, in: Handbook of Heterogeneous Catalysis, Vol. 5, eds. G. Ertl, H. Knözinger and J. Weitkamp (VCH, Weinheim, 1997) p. 2334.
M.G. Turcotte and T.A. Johnson, in: Encyclopedia of Chemical Technology, Vol. 2, eds. F.M. Mark, D.F. Othmer, C.G. Overberger and G.T. Seaborg (Wiley, New York, 1992) p. 369.
P.F. Vogt and J.J. Gerulis, in: Ullmann's Encyclopedia of Industrial Chemistry, Vol. A2, ed. B. Elvers (VCH, Weinheim, 1989) p. 37.
M. Deeba, M.E. Ford and T.A. Johnson, in: Catalysis of Organic Reactions, ed. D.W. Blackburn (Dekker, New York, 1990) p. 241.
D.M. Roundhill, Chem. Rev. 92 (1992) 1.
V. Zamlynny, L. Kubelková, E. Baburek, K. Jirátová and J. Nováková, Appl. Catal. A 169 (1998) 119.
A. Fischer, T. Mallat and A. Baiker, Angew. Chem. Int. Ed. Eng. 38 (1999) 351.
A. Fischer, M. Maciejewski, T. Bürgi, T. Mallat and A. Baiker, J. Catal. 183 (1999) 373.
A. Fischer, T. Mallat and A. Baiker, J. Catal. 182 (1999) 289.
A. Fischer, T. Mallat and A. Baiker, J. Mol. Catal., in press.
L.W. Watts, Jr. and H.G. Waddill, US Patent 4181682 (1980), to Texaco Development Corp.
Y. Ichikawa, E. Yoshisato and K. Nakagawa, US Patent 4151204 (1979), to Teijin Limited.
A. Baiker and W. Richarz, Ind. Eng. Chem. Prod. Res. Dev. 16 (1977) 261.
G.A. Gibson and J.R. Winters, US Patent 4400539 (1983), to Union Carbide Corporation.
J.W. Labadie and D.D. Dixon, J. Mol. Catal. 42 (1987) 367.
G. Sirokmán, Á. Molnár and M. Bartók, J. Mol. Catal. 19 (1983) 35.
J.C. Giddings, M.N. Myers, L. McLaren and R.A. Keller, Science 162 (1968) 67.
A. Baiker, Chem. Rev. 99 (1999) 453.
J.M.H. Levelt Sengers, in: Supercritical Fluids — Fundamentals for Application, eds. E. Kiran and J.M.H. Levelt Sengers (Kluwer, Dordrecht, 1994) p. 3.
P.E. Savage, S. Gopalan, T.I. Mizan, C.J. Martino and E.E. Brock, AIChE J. 41 (1995) 1723.
K.P. Johnston and C. Haynes, AIChE J. 33 (1987) 2017.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Jenzer, G., Mallat, T. & Baiker, A. Cobalt‐catalyzed amination of 1,3‐cyclohexanediol and 2,4‐pentanediol in supercritical ammonia. Catalysis Letters 61, 111–114 (1999). https://doi.org/10.1023/A:1019045527193
Issue Date:
DOI: https://doi.org/10.1023/A:1019045527193