Abstract
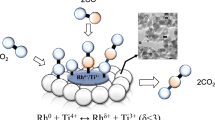
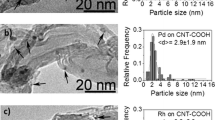
Carbon nanotubes (CNTs) and CNTs-supported rhodium were tested as catalysts for NO decomposition. For the fresh catalysts, 100% NO conversion was achieved at 600°C over CNTs; when 1 wt% Rh was loaded on CNTs, 100% NO conversion was achieved at 450°C. If the catalysts were pre-reduced in H2 at or above 300°C, 100% NO conversions were observed at 300°C. XPS investigation indicated that there was still metallic rhodium (BE=307.2 eV) on Rh/CNTs after heating in air at 500°C for 2 h and after the NO decomposition reaction. As for a 1 wt% Rh/Al2O3 sample, the rhodium (BE = 308.2 eV) was completely in the form of Rh2O3 after similar treatments. These results suggest that compared to γ-Al2O3, the CNTs material is more capable of keeping the rhodium in its metallic state. The results obtained in H2-TPR studies support this conclusion. In addition, TEM investigation revealed that the rhodium particles distributed rather evenly over CNTs with a particle diameter of around 8 nm. We propose that CNTs can be used as a material for the facilitation of NO decomposition.
Similar content being viewed by others
References
M.S. Dresselhaus, G. Dresselhaus and P.C. Eklund, eds., Science of Fullerenes and Carbon Nanotubes (Academic Press, London, 1995).
A.C. Dillon, K.M. Jones, T.A. Bekkedahl, C.H. Klang, D.S. Bethane and M.I. Heben, Nature (London) 386 (1997) 377.
P. Chen, X. Wu, J. Lin and K.L. Tan, Science 285 (1999) 91.
B. Coq, J.M. Planeix and V. Brotons, Appl. Catal. A 173 (1998) 175.
J.M. Planeix, N. Coustel, B. Coq, V. Brotons, P.S. Kumbhar, R. Dutartre, P. Geneste, P. Bernier and P.M. Ajayan, J. Am. Chem. Soc. 116 (1994) 7935.
Th. Braun, M. Wohlers, T. Belz, G. Nowizke, G. Wortmann, Y. Uchida, N. Pfänder and R. Schlögl, Catal. Lett. 43 (1997) 167.
Th. Braun, M. Wohlers, T. Belz and R. Schlögl, Catal. Lett. 43 (1997) 175.
J.C. Calvert, Pure Appl. Chem. 69 (1997) 1.
V.I. Parvulescu, P. Grange and B. Delmon, Catal. Today 46 (1998) 233.
M. Iwamoto, H. Furukawa, Y. Mine, F. Uemura, S. Mikuriya and S. Kagawa, J. Chem. Soc. Chem. Commun. (1986) 1272.
A. Gervasini, P. Carniti and V. Ragaini, Appl. Catal. B 22 (1999) 201.
A.M. Pisanu and C.E. Gigola, Appl. Catal. B 20 (1999) 179.
S. Oh and C. Eickel, J. Catal. 128 (1991) 256.
B. Cho, B. Shanks and J. Bailey, J. Catal. 115 (1989) 486.
B. Cho, J. Catal. 148 (1994) 697.
A.M. Pisanu and C.E. Gigola, Appl. Catal. B 20 (1999) 179.
J.Z. Luo, Z.L. Yu, C.F. Ng and C.T. Au, J. Catal., submitted.
K. Tohji, H. Takahashi, Y. Shinoda, N. Shimizu, B. Jeyadevon, I. Matsuoka, Y. Saito, A. Kasuya, S. Ito and Y. Nishina, J. Phys. Chem. B 101 (1997) 1974.
Handbook of X-Ray Photoelectron Spectroscopy (Perkin-Elmer, Eden Prairie, MN).
L.Z. Gao and C.T. Au, Catal. Lett. 65 (2000) 91.
M. Rasinkangas, T.T. Pakkanen and T.A. Pakkanen, J. Organomet. Chem. 476 (1994) C6.
M.J. Illán-Gómez, E. Raymundo-Pinero, A. García-García, A. Linares-Solano and C. Salinas-Martinez de Lecea, Appl. Catal. B 20 (1999) 267.
A. García-García, M.J. Illán-Gómez, E. Raymundo-Pinero, A. Linares-Solano and C. Salinas-Martinez de Lecea, Appl. Catal. B 25 (2000) 39.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Luo, J., Gao, L., Leung, Y. et al. The decomposition of NO on CNTs and 1 wt% Rh/CNTs. Catalysis Letters 66, 91–97 (2000). https://doi.org/10.1023/A:1019035220233
Issue Date:
DOI: https://doi.org/10.1023/A:1019035220233