Abstract
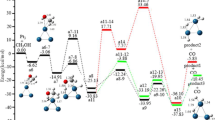
Theoretical studies of CO hydrogenation to methanol over Cu, Pd, and Pt metals have been carried out using a quasi‐relativistic density‐functional method. The metal surface is simulated by a M10 cluster model. Reaction energies for the elementary steps involved are determined. The activation energies are estimated by the analytic BOC‐MP formula. The results support that these metals are active in CO hydrogenation to methanol. The rate‐determining steps are shown to be different for the metals. The highest activation energies of reaction on the metals fall in the order Cu < Pd < Pt, which corresponds to the order of the catalytic activities of the metals in CO hydrogenation.
Similar content being viewed by others
References
T.H. Fleisch and R.L. Mieviller, J. Catal. 90 (1984) 165.
G.C. Chinchen, P.J. Denny, J.R. Jennings, M.S. Spencer and K.C. Waugh, Appl. Catal. 36 (1988) 1.
M.L. Poutsma, L.F. Elek, P.A. Ibarbia, A.P. Risch and J.A. Rabo, J. Catal. 52 (1978) 157.
A. Takeuchi and R. Katzer, J. Phys. Chem. 86 (1982) 2438.
J.M. Thomas and W.J. Thomas, Principles and Practice of Heterogeneous Catalysis (VCH, Weinheim, 1997) ch. 8.
F. Fischer and H. Tropsch, Brennst. Chem. 7 (1926) 97.
R.W. Joyner, J. Catal. 50 (1977) 176.
A. Jones and B.D. McNicol, J. Catal. 47 (1977) 384.
M. Araki and V. Ponec, J. Catal. 44 (1976) 439.
P. Biloen, J.N. Helle and W.M.H. Sachtler, J. Catal. 58 (1979) 95.
Y. Kikuzono, S. Kagami, S. Naito, T. Onishi and K. Tamaru, Faraday Disc. Chem. Soc. 72 (1981) 143.
A. Takeuchi and J.R. Katzer, J. Phys. Chem. 85 (1981) 937.
E. Shustorovich and A.T. Bell, J. Catal. 113 (1988) 341; A.T. Bell and E. Shustorovich, J. Catal. 121 (1990) 1.
R.D. Srivastava, Heterogeneous Catalytic Science (CRC Press, Boca Raton, FL, 1988) ch. IV.
R.G. Herman, K. Klier, G.W. Simmons, B.P. Finn, J.B. Bulko and T.P. Kobylinski, J. Catal. 56 (1979) 407; S. Mehta, G.W. Simmons, K. Klier and R.G. Herman, J. Catal. 57 (1979) 339; J.B. Bulko, R.G. Herman, K. Klier and G.W. Simmons, J. Phys. Chem. 83 (1979) 3118.
S.P.S. Andrew, Post Congr. Symp. Int. Congr. Catal. 7th Osakar (1980).
G.C. Chinchen, K.C. Waugh and D.A. Whan, Appl. Catal. 25 (1986) 101; G.C. Chinchen, C.M. Hay, H.D. Vandervell and K.C. Waugh, J. Catal. 103 (1987) 79.
B.S. Clausen, G. Steffensen, B. Fabius, J. Villadsen, R. Feidenhaus'l and H. Topsøe, J. Catal. 132 (1991) 524.
J.B. Friedrich, M.S. Wainwright and D.J. Young, J. Catal. 80 (1983) 1.
E.J. Baerends, D.E. Ellis and P. Ros, Chem. Phys. 2 (1973) 41; G. te Velde and E.J. Baerends, J. Comp. Phys. 99 (1992) 84.
S.H. Vosko, L. Wilk and M. Nusair, Canad. J. Phys. 58 (1980) 1200.
A.D. Becke, Phys. Rev. A 38 (1988) 3098.
J.P. Perdew, Phys. Rev. B 33 (1986) 8822.
T. Ziegler, V. Tschinke, E.J. Baerends, J.G. Snijders and W. Ravenek, J. Phys. Chem. 93 (1989) 3050.
C.T. Au, M.S. Liao and C.F. Ng, J. Phys. Chem. 102 (1998) 3959.
E. Shustorovich, Adv. Catal. 37 (1990) 101.
T.N. Rhodin and G. Ertl, The Nature of the Surface Chemical Bond (North-Holland, Amsterdam, 1979) ch. 5.
G.E. Gdowski, J.A. Fair and R.J. Madix, Surf. Sci. 127 (1983) 541.
P. Hollins and J. Pritchard, Surf. Sci. 127 (1983) 541.
R.J. Behm, K. Christmann, G. Ertl and M.A. van Hove, J. Chem. Phys. 73 (1980) 2984.
E. Schweizer, B.N.J. Persson, M. Tushaus, D. Hoge and A.M. Bradshaw, Surf. Sci. 213 (1989) 49.
C. Zheng, Y. Apeloig and R. Hoffmann, J. Am. Chem. Soc. 110 (1988) 749.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Au, C., Zhou, T. & Lai, W. Theoretical studies of CO hydrogenation to methanol over Cu, Pd, and Pt metals. Catalysis Letters 62, 147–151 (1999). https://doi.org/10.1023/A:1019019710780
Issue Date:
DOI: https://doi.org/10.1023/A:1019019710780