Abstract
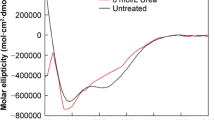
A critical problem in the storage and delivery of pharmaceutical proteins is aggregation in the solid state induced by elevated temperature and moisture. These conditions are particularly relevant for studies of protein stability during accelerated storage or for proteins loaded in polymeric delivery devices in vivo. In the present investigation, we have found that, when exposed to an environment simulating these conditions, lyophilized insulin undergoes both covalent and noncovalent aggregation. The covalent process has been elucidated to be intermolecular thiol-catalyzed disulfide interchange following β-elimination of an intact disulfide bridge in the insulin molecule. This process is accelerated by increasing the temperature and water content of the insulin powder or by performing lyophilization and/or dissolution of insulin in alkaline media. The aggregation can be ameliorated by the presence of Cu 2+ , which presumably catalyzes the oxidization of free thiols. The water sorption isotherm for insulin reveals that the extent of aggregation directly correlates with the water uptake by the lyophilized insulin powder, thus pointing to the critical role of protein conformational mobility in the aggregation process.
Similar content being viewed by others
REFERENCES
M. Thayer. Biopharmaceuticals overcoming market hurdles. Chem. Eng. News 69:27–48 (1991).
M. C. Manning, K. Patel, and R. T. Borchardt. Stability of protein pharmaceuticals. Pharm. Res. 6:903–918 (1989).
M. J. Hageman. The role of moisture in protein stability. Drug Dev. Ind. Pharm. 14:2047–2070 (1988).
R. Pearlman and T. Nguyen. Pharmaceutics of protein drugs. J. Pharm. Pharmacol. 44:179–185 (1992).
J. Brange. Galenics of Insulin: The Physico-chemical and Pharmaceutical Aspects of Insulin and Insulin Preparations. Springer-Verlag, Berlin, 1987.
L. Brown, C. Munoz, L. Siemer, E. Edelman, and R. Langer. Controlled release of insulin from polymer matrices. Control of diabetes in rats. Diabetes 35:692–697 (1986).
A. Rupley, E. Gratton, and G. Careri. Water and globular proteins. Trends Biochem. Sci. 8:18–22 (1983).
D. B. Volkin, A. Staubli, R. Langer, and A. M. Klibanov. Enzyme thermoinactivation in anhydrous organic solvents. Biotechnol. Bioeng. 37:843–853 (1991).
K. A. Conners, G. L. Amidon, and V. J. Stella. Chemical Stability of Pharmaceuticals, Wiley, New York, 1986, Chap. 6.
R. Liu, R. Langer, and A. M. Klibanov. Moisture-induced aggregation of lyophilized proteins in the solid state. Biotechnol. Bioeng. 37:177–184 (1991).
C. C. Hsu, C. A. Ward, R. Pearlman, H. M. Nguyen, D. A. Yeung, and J. G. Curley. Determining the optimum residual moisture in lyophilized protein pharmaceuticals. Dev. Biol. Stand. 74:255–271 (1991).
M. J. Hageman, J. M. Bauer, P. L. Possert, and R. T. Darrington. Preformulation studies oriented toward sustained delivery of recombinant somatotropins. J. Agr. Food Chem. 40:348–355 (1992).
P. W. Riddles, R. L. Blakeley, and B. Zerner. Reassessment of Ellman's reagent. Meth. Enzymol. 91:49–60 (1983).
A. Conners. The Karl Fischer titration of water. Drug Dev. Ind. Pharm. 14:1891–1903 (1988).
M. J. Hageman, P. L. Possert, and J. M. Bauer. Prediction and characterization of the water sorption isotherm for bovine somatotropin. J. Agr. Food Chem. 40:342–347 (1992).
J. F. Youngs. Humidity control in the laboratory using salt solutions—a review. J. Appl. Chem. 17:241–245 (1967).
L. Greenspan. Humidity fixed points of binary saturated aqueous solutions. J. Res. NBS 81A:89–96 (1977).
S. Brunauer, P. H. Emmett, and E. Teller. Adsorption of gases in multimolecular layers. J. Am. Chem. Soc. 60:309–319 (1938).
M. Schnepf. Protein-water interactions. In T. M. I. Hardman (ed.), Water and Food Quality, Elsevier, New York, 1989, pp. 135–168.
B. K. Milthorpe, L. W. Nichol, and P. D. Jeffrey. The polymerization pattern of Zn(II)-insulin at pH 7.0. Biochim. Biophys. Acta 495:195–202 (1977).
H. Thurow and K. Geisen. Stabilization of dissolved proteins against denaturation at hydrophobic interfaces. Diabetologia 27:212–218 (1984).
V. Sluzky, J. A. Tamada, A. M. Klibanov, and R. Langer. Kinetics of insulin aggregation in aqueous solutions upon agitation in the presence of hydrophobic surfaces. Proc. Natl. Acad. Sci. USA 88:9377–9381 (1991).
J. Brange, S. Havelund, and P. Hougaard. Chemical stability of insulin. 2. Formation of higher molecular weight transformation products during storage of pharmaceutical preparations. Pharm. Res. 9:727–734 (1992).
J. Brange and L. Langkjaer. Chemical stability of insulin. 3. Influence of excipients, formulation, and pH. Acta Pharm. Nord. 4:149–158 (1992).
J. Brange. Chemical stability of insulin. 4. Mechanisms and kinetics of chemical transformations in pharmaceutical formulation. Acta Pharm. Nord. 4:209–222 (1992).
B. V. Fisher and P. B. Porter. Stability of bovine insulin. Pharm. Pharmacol. 33:203–206 (1980).
E. Helmerhorst and G. B. Stokes. Generation of an acid-stable and protein-bound persulfide residue in alkali-or sulfhydryl-treated insulin by a mechanism consonant with the β-elimination hypothesis of disulfide bond lysis. Biochemistry 22:69–75 (1983).
A. M. Klibanov. Enzyme catalysis in anhydrous organic solvents. Trends Biochem. Sci. 14:141–144 (1989).
J. D. Hopton, C. J. Swan, and D. L. Trimm. Liquid-phase oxidation of thiols to disulfides. Adv. Chem. Ser. 75:216–224 (1968).
L. Pauling. The absorption of water by proteins. J. Am. Chem. Soc. 67:555–557 (1945).
R. W. Green. The adsorption of water vapor on casein. Trans. Roy. Soc. N.Z. 77:313–317 (1948).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Costantino, H.R., Langer, R. & Klibanov, A.M. Moisture-Induced Aggregation of Lyophilized Insulin. Pharm Res 11, 21–29 (1994). https://doi.org/10.1023/A:1018981208076
Issue Date:
DOI: https://doi.org/10.1023/A:1018981208076