Abstract
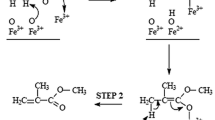
The transesterification of methylmethacrylate with 1-dodecanol has been studied by using Brønsted acid and Lewis acid type catalysts either for homogeneous or heterogeneous systems. Initial rates studies in a batch reactor lead to a greater efficiency for the Lewis acid catalysts (soluble or supported titanates). The activity of the supported catalyst can be linked to the molecular association of the titanates during the fixation on the silica support, the existence of monomeric species corresponding to the most important activity. The nature of the support has also an influence: for the same density of titanium atoms, a smaller surface area leads to a better efficiency.
Similar content being viewed by others
References
E. Nasr, R. Guiraud, J.-L. Pellegatta and C. Blandy, Can. J. Chem. Eng. 73 (1995) 129.
C. Blandy, J.-L. Pellegatta, R. Choukroun, B. Gilot and R.Guiraud, Can. J. Chem. 71 (1993) 34.
C. Blandy, J.-L. Pellegatta and B.Gilot, J. Catal. 150 (1994) 150.
H.A. Benesi and B.H.C. Winguist, in: Advances in Catalysis, Vol. 27, eds. D.D. Eley, H. Pines and P.B. Weiz (Academic Press, New York, 1978) p. 97.
M. Marsi, Inorg. Chem. 27 (1988) 3067.
A. Gicquel and B. Torck, J. Catal. 83 (1983) 9.
C. Blandy, R. Guerreiro and D. Gervais, J. Organomet. Chem. 128 (1977) 415.
M.D. Rausch and H.B. Gordon, J. Organomet. Chem. 74 (1974) 85.
Rights and permissions
About this article
Cite this article
Blandy, C., Pellegatta, JL. & Cassoux, P. Homogeneous and supported titanates as catalysts for transesterification of acrylic esters. Catalysis Letters 43, 139–142 (1997). https://doi.org/10.1023/A:1018978203665
Issue Date:
DOI: https://doi.org/10.1023/A:1018978203665