Abstract
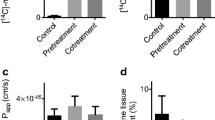
The aim of this study was to characterize transport of FITC-labeled dextrans of different molecular weights as model compounds for peptides and proteins through buccal mucosa. The penetration of these dextrans through porcine buccal mucosa (a nonkeratinized epithelium, comparable to human buccal mucosa) was investigated by measuring transbuccal fluxes and by analyzing the distribution of the fluorescent probe in the epithelium, using confocal laser scanning microscopy for visualizing permeation pathways. The results revealed that passage of porcine buccal epithelium by hydrophilic compounds such as the FITC-dextrans is restricted to permeants with a molecular weight lower than 20 kDa. The permeabilities of buccal mucosa for the 4- and 10-kDa FITC-dextran (of the order of 10−8 cm/sec) were not significantly different from each other or from the much smaller compound FITC. The confocal images of the distribution pattern of FITC-dextrans showed that the paracellular route is the major pathway through buccal epithelium.
Similar content being viewed by others
REFERENCES
V. H. L. Lee. Enzymatic barriers to peptide and protein absorption. Crit. Rev. Ther. Drug Carrier Syst. 5:69–97 (1988).
J. C. Verhoef, H. E. Boddé, A. G. de Boer, J. A. Bouwstra, H. E. Junginger, F. W. H. M. Merkus, and D. D. Breimer. Transport of peptide and protein drugs across biological membranes. Eur. J. Drug Metab. Pharmacokinet. 15:83–93 (1990).
M. E. de Vries, H. E. Boddé, J. C. Verhoef, and H. E. Junginger. Developments in buccal drug delivery. Crit. Rev. Ther. Drug Carrier Syst. 8:271–303 (1991).
N. F. H. Ho, C. L. Barsuhn, P. S. Burton, and H. P. Merkle. Routes of delivery: Case studies, (3) Mechanistic insights to buccal delivery of proteinaceous substances. Adv. Drug Del. Rev. 8:197–235 (1992).
H. P. Merkle and G. Wolany. Buccal delivery for peptide drugs. J. Control. Rel. 21:155–164 (1992).
C. A. Squier. The permeability of oral mucosa. Crit. Rev. Oral Biol. Med. 2:13–32 (1991).
M. E. de Vries, H. E. Boddé, J. C. Verhoef, M. Ponec, W. I. H. M. Craane, and H. E. Junginger. Localization of the permeability barrier inside porcine buccal mucosa: A combined in vitro study of drug permeability, electrical resistance and tissue morphology. Int. J. Pharm. 76:25–35 (1991).
I. A. Siegel. Permeability of the oral mucosa. In J. Meyer, C. A. Squier, and S. J. Gerson (eds.), The Structure and Function of Oral Mucosa, Pergamon Press, London, 1984, pp. 95–108.
R. O. Potts and R. H. Guy. Predicting skin permeability. Pharm. Res. 9:663–669 (1992).
C. A. Lesch, C. A. Squier, A. Cruchley, D. M. Williams, and P. Speight. The permeability of human oral mucosa and skin to water. J. Dent. Res. 68:1345–1349 (1989).
P. W. Wertz and C. A. Squier. Cellular and molecular basis of barrier function in oral epithelium. Crit. Rev. Ther. Drug Carrier Syst. 8:237–269 (1991).
D. M. Shotton. Confocal scanning optical microscopy and its applications for biological specimens. J. Cell Sci. 94:175–206 (1989).
C. Cullander and R. H. Guy. Visualizing the pathways of iontophoretic current flow in real time with laser-scanning confocal microscopy and the vibrating probe electrode. In R. C. Scott, R. H. Guy, J. Hadgraft, and H. E. Boddé (eds.), Prediction of Percutaneous Penetration, Methods, Measurements, Modelling, Vol. 2, IBC Technical Services, London, 1991, pp. 229–237.
D. Harris and J. R. Robinson. Effects of inflammation on buccal permeability. Proc. Int. Symp. Control. Rel. Bioact. Mater. 17:228–229 (1990).
M. E. Dowty, K. E. Knuth, B. K. Irons, and J. R. Robinson. Transport of thyrotropin releasing hormone in rabbit buccal mucosa in vitro. Pharm. Res. 9:1113–1122 (1992).
E. Quadros, J. Cassidy, K. Gniecko, and S. LeRoy. Buccal and colonic absorption of CGS 16617, a novel ACE inhibitor. J. Control. Rel. 19:77–86 (1991).
S. Senel, A. J. Hoogstraate, F. Spies, H. E. Junginger, and H. E. Boddé. Enhancing effects of bile salts on buccal penetration of fluorescent markers: Kinetic and histological studies. Pharm. Weekblad Sci. Ed. F36 Suppl. F:54 (1992).
S.-Y. Chen and C. A. Squier. The ultrastructure of the oral epithelium. In J. Meyer, C. A. Squier, and S. J. Gerson (eds.), The Structure and Function of Oral Mucosa, Pergamon Press, London, 1984, pp. 7–30.
N. Uchida, Y. Maitani, Y. Machida, M. Nakagaki, and T. Nagai. Influence of bile salts on the permeability of insulin through the nasal mucosa of rabbits in comparison with dextran derivatives. Int. J. Pharm. 74:95–103 (1991).
T. Ohtani, M. Murakami, A. Yamamoto, K. Takada, and S. Muranishi. Effect of absorption enhancers on pulmonary absorption to fluorescein isothiocyanate dextrans with various molecular weights. Int. J. Pharm. 77:141–150 (1991).
J. B. M. M. van Bree, A. G. de Boer, M. Danhof, L. A. Ginsel, and D. D. Breimer. Characterization of an “in vitro” blood brain barrier (BBB): Effects of molecular size and lipophilicity on cerebrovascular endothelial transport rates of drugs. J. Pharmacol. Exp. Ther. 247:1233–1239 (1988).
C. A. Squier and C. A. Lesch. Penetration pathways of different compounds through epidermis and oral epithelium. J. Oral Pathol. 17:512–516 (1988).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Hoogstraate, A.J., Cullander, C., Nagelkerke, J.F. et al. Diffusion Rates and Transport Pathways of Fluorescein Isothiocyanate (FITC)-Labeled Model Compounds Through Buccal Epithelium. Pharm Res 11, 83–89 (1994). https://doi.org/10.1023/A:1018949828548
Issue Date:
DOI: https://doi.org/10.1023/A:1018949828548