Abstract
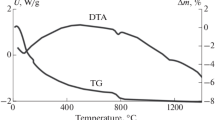
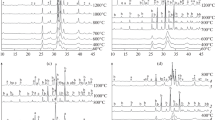
Apatites were prepared with three different fluoride concentrations: 0.0 mM (pure hydroxyapatite) 2.5 mM and 5 mM. Reactions were performed in aqueous medium using a reaction between diammonium orthophosphate and calcium nitrate 4-hydrate and ammonium fluoride at temperatures of 3°, 25°, 60° and 90°C. The effects of reaction temperature and fluoride concentration on the crystal morphology, phase purity and crystallinity of the precipitates were observed, using transmission electron microscopy (TEM), X-ray diffraction (XRD), Fourier transform infrared (FTIR) spectroscopy and ion chromatography. Transmission electron micrographs revealed that the crystallites precipitated at 3°C were spheroidal, but became increasingly acicular with increasing precipitation temperature. X-ray diffraction results indicated that all the materials produced were phase pure and that the crystallinity of apatites prepared at higher precipitation temperatures was higher than those prepared at lower precipitation temperatures. A significant difference in the a-axis dimension of fluoride-substituted apatites was observed, as compared to hydroxyapatite. FTIR spectroscopy revealed a hydroxyl band at 3568 cm-1, along with a broad peak of adsorbed water in the region of 3568 cm-1 to 2670 cm-1 in the hydroxyapatite and fluoride-substituted apatite powders. Hence by careful selection of the precipitation conditions and fluoride contents, the composition and morphology of fluoride-substituted apatite may be controlled and this has interesting implications for the development of these materials for biomedical implantation.
Similar content being viewed by others
References
F. BETTS, C. NORMAN and S. R. POSNER, J. Cryst. Growth 53 (1981) 63.
I. ARENDS I and I. M. TENCATE, ibdi. 53 (1981) 135.
G. BOIVIN and P. J. MEUNIER, in ‘‘The metabollic and molecular basis of acquired disease’’, edited by R. D. Cohen, B. Lewis, K. G. M. M. Alberti and A. M. Desman (Bailliere Tindall, London, 1990).
W. G. M. VAN DEN HOCK, T. P. FENSTRA and P. L. deBRUYN, J. Phys. Chem. 84 (1980) 3312.
R. Z. LeGEROS, R. KIJKOWSKA, W. JIA and J. P. LeGEROS, J. Fluorine Chem. 41 (1988) 53–64.
J. P. BARONE and G. H. NANCOLLAS, J. Dent. Res. 57 (1978) 735–742.
E. C. MORENO, M. KREWASK, R. T. ZAHRADINK, Nature 247 (1974) 64.
G. H. NANCOLLAS, Z. AHMED and P. KOUTSOUKOS, in ‘‘ACS Symposium Series’’, Vol. 93, pp. 475–497 (American Chemical Society, Washington, DC, 1979).
E. C. MORENO and K. VARUGHESE, J. Cryst. Growth 53 (1981) 20.
P. PANTUMVANIT, F. F. FEAGIN and T. KOULOURIDES, Caries Res. 11 (1977) 52.
I. L. MEYER and G. H. NANCOLLAS, J. Dent. Res. 51 (1972) 1443.
K. ISHIKAWA, E. D. EANES and M. S. TUNG, ibid. 73 (1994) 1462.
E. DUFF, J. Chem. Soc. A, (1971) 33.
F. C. M. DRIESSENS, in ‘‘Bioceramics of calcium phosphate’’, edited by K. deGroot (CRC Press, Boca Raton, FL, 1988), pp. 1–33.
J. CHRISTOFFERSEN, M. R. CHRISTOFFERSEN and T. JOHANSEN, in 12th European Conference of Biomaterials, Porto, Portugal, 1995.
J. CHRISTOFFERSEN, J. Cryst. Growth 49 (1980) 29.
M. R. CHRISTOFFERSEN, J. CHRISTOFFERSEN and W. KIBALCZYC, ibid. 106 (1990) 349.
M. R. CHRISTOFFERSEN and J. CHRISTOFFERSEN, ibid. 121 (1990) 349.
K. S. TENHUISEN and P. W. BROWN, J. Dent. Res. 73 (1994) 598.
K. KANDORI, A. YASUKAWA and T. ISHIKAWA, Chem. Mater. 7 (1995) 26.
K. ISHIKAWA, E. D. EANES and M. S. TUNG, J. Dent. Res. 73 (1994) 1462.
S. V. DOROZHKIN, Russian J. Inorg. Chem. 39 (1994) 217.
A. C. LARSON, R. B. Von DREELE and M. LUJAN Jr., ‘‘GSAS-Generalised Crystal Structure Analysis System’’. Neutron Scattering Centre, Los Alamos National Laboratory, California (1990).
I. ABRAHAMS and J. C. KNOWLES, J. Mater. Chem. 4 (1994) 185.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
JHA , L.J., BEST , S.M., KNOWLES , J.C. et al. Preparation and characterization of fluoride-substituted apatites. Journal of Materials Science: Materials in Medicine 8, 185–191 (1997). https://doi.org/10.1023/A:1018531505484
Issue Date:
DOI: https://doi.org/10.1023/A:1018531505484