Abstract
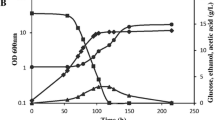
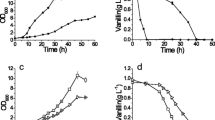
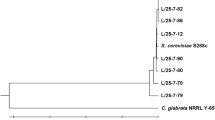
Three strains of Saccharomyces cerevisiae, SC-1, DBVPG 6173 and DBVPG 6037, were studied for vanadate resistance in complex Sabouraud medium since they did not thrive in different minimal media (yeast nitrogen base with and without amino acids). The strain SC-1 was resistant up to 16 mm of vanadate, whereas the strains DBVPG 6173 and DBVPG 6037 were inhibited by 8 mm and 4 mm vanadate, respectively. The vanadate resistance in strain SC-1 was constitutive and due to the reduction of this oxyanion to vanadyl, which was detected by EPR spectroscopy and visible spectroscopy. The transformation of vanadate to vanadyl took place during the exponential growth phase; 10 mm of vanadate was reduced to vanadyl outside the cells since the oxyanion was not detected in the cell biomass and only a negligible concentration of vanadyl (25 nmoles mg cells dry weight) was found in the biomass. The other two vanadate-sensitive yeast strains only accumulated vanadate and did not reduce the oxyanion to vanadyl.
Similar content being viewed by others
References
Armstrong EM, Calviou LJ, Charnock JMet al. 1991 Studies on amavadin and related chemical systems. J Inorg Biochem 43, 413.
Bowman BJ. 1983 Vanadate uptake inNeurospora crassaoccurs via phosphate transport system II. J Bacteriol 153, 286–291.
Bowman BJ, Slayman CW. 1979 The effects of vanadate on the plasma membrane ATPase ofNeurospora crassa. J Biol Chem 254, 2928–2934.
Butler A, Soedjak HS, Tschirret-Guth RA. 1991 Chlorination catalyzed by vanadium. J Inorg Biochem 43, 405.
Cantley LC Jr, Aisen P. 1979 The fate of cytoplasmic vanadium. J Biol Chem 254, 1781–1784.
Cantley LC Jr, Josephson L, Warner Ret al. 1977 Vanadate is a potent (Na,K)-ATPase inhibitor found in ATP derived from muscle. J Biol Chem 252, 7421–7423.
Cantley LC Jr, Cantley LG, and Josephson L. 1978a A characterization of vanadate interaction with the (Na,K)ATPase. J Biol Chem 253, 7361–7368.
Cantley LC Jr, Resh MD, Guidotti G. 1978b Vanadate inhibits the red cell (Na+,K+)ATPase from the cytoplasmic side. Nature 272, 552–554.
De Boer E, Tromp MGM, Krenn GE, Wever R. 1986a Vanadium(V) as an essential element for haloperoxidase activity in marine brown algae: purification and characterization of a vanadium(V)-containing bromoperoxidase fromLaminaria saccharina. Biochim Biophys Acta 872, 104–115.
De Boer E, van Kooyk Y, Tromp MGM, Plat H, Wever R. 1986b Bromoperoxidase fromAscophyllum nodosum: a novel class of enzymes containing vanadium as a prosthetic group?Biochim Biophys Acta 869, 48–53.
Fallik E, Hartel PG, Robson RL. 1993 Presence of a vanadium nitrogenase inAzotobacter paspali. Appl Environ Microbiol 59, 1883–1886.
Gresser MJ, Tracey AS. 1990 Vanadate as phosphate analog in biochemistry. In: Chasteen N. Dennis (ed)Vanadium in Biological Systems. Dordrecht: Kluwer Academic Publishers; 63–79.
Hales BJ, Case EE, Morningstar JE, Dzeda MF, Mauterer LA. 1986 Isolation of a new vanadium-containing nitrogenase fromAzotabacter vinelandii. Biochemistry 25, 7251–7255.
Hawkins CJ. 1991 Vanadium in theAscidiacea. J Inorg Biochem 43, 402.
Lyalikova NN, Yurkova NA. 1992 Role of microorganisms in vanadium concentration and dispersion. Geomicrobiol J 1015–26.
Macara IG. 1980 Vanadium-an element in search of a role. Trends Biochem Sci 4, 92–94.
Michibata H, Hirose H, Sugiyama K, Ookubo Y, Kanamori K. 1990 Extraction of a vanadium-binding substance (vanadobin) from the blood cells of several ascidian species. Biol Bull 179, 140–147.
Minasi LA, Willsky GR. 1991 Characterization of vanadate-dependent NADH oxidation stimulated bySaccharomyces cerevisiaeplasma membranes. J Bacteriol 173, 834–841.
Minasi LA, Chang A, Willsky GR. 1990 Plasma membrane-stimulated vanadate-dependent NADH oxidation is not the primary mediator of vanadate toxicity inSaccharomyces cerevisiae. J Biol Chem 265, 14907–14910.
Rehder D. 1991 The bioinorganic chemistry of vanadium. Angew Chem Int Ed Engl 30, 148–167.
Rehder D. 1992 Structure and function of vanadium compounds in living organisms. Biometals 5, 3–12.
Rehder D, Priebsch W, Holst H, Hillerns F, Richter J. 1991 (Model) investigations of the function of vanadium in biological system. J Inorg Biochem 43, 409.
Robson RL, Eady RR, Richardson TH, et al. 1986 The alternative nitrogenase ofAzotobacter chroococcumis a vanadium enzyme. Nature 322, 388–390.
Silver S, Walderhaug M. 1992 Gene regulation of plasmidand chromosome-determined inorganic ion transport in bacteria. Microbiol Rev 56, 195–228.
Simons TJB. 1979 Vanadate-a new tool for biologists. Nature 281, 337–338.
Singh HN, Chakravarty D, Srinivasa Rao K, Singh AK. 1993 Vanadium requirement for growth on N2 or nitrate as nitrogen source in a tungsten-resistant mutant of the cyanobacteriumNostoc muscorum. J Basic Microbiol 33, 201–205.
Wever R, Tromp MGM. 1991 Structure and function of vanadium bromoperoxidases. J Inorg Biochem 43, 404.
Willsky GR. 1979 Characterization of the plasma membrane Mg2+-ATPase from the yeast, Saccharomyces cerevisiae. J Biol Chem 254, 3326–3332.
Willsky GR, White DA, McCabe BC. 1984 Metabolism of added orthovanadate to vanadyl and high-molecularweight vanadates bySaccharomyces cerevisiae. J Biol Chem 259, 13273–13281.
Willsky GR, Leung JO, Offerman PV Jr, Plotnik EK, Dosch SF. 1985 Isolation and characterization of vanadate-resistant mutants ofSaccharomyces cerevisiae. J Bacteriol 164, 611–617.
Zoroddu MA, Fruianu M, Dallocchio R, Masia A. 1996. Electron paramagnetic resonance studies and effects of vanadium inSaccharomyces cerevisiae. BioMetals 9, 91–97.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bisconti, L., Pepi1, M., Mangani, S. et al. Reduction of vanadate to vanadyl by a strain of Saccharomyces cerevisiae. Biometals 10, 239–246 (1997). https://doi.org/10.1023/A:1018360029898
Issue Date:
DOI: https://doi.org/10.1023/A:1018360029898