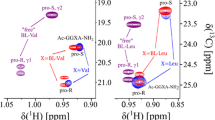
Abstract
The effects of the commonly used denaturant guanidine hydrochloride(GuHCl) on the random coil conformations and NMR chemical shifts of theproteogenic amino acids have been characterized using the peptide seriesAc-Gly-Gly-X-Gly-Gly-NH2. The φ angle-sensitive couplingconstants, ROESY cross peak intensities and proline cis–trans isomerratios of a representative subset of these peptides are unaffected by GuHCl,which suggests that the denaturant does not significantly perturb intrinsicbackbone conformational preferences. A set of3JHNHα values is presented which agreewell with predictions of recently developed models of the random coil. Wehave also measured the chemical shifts of all 20 proteogenic amino acids inthese peptides over a range of GuHCl concentrations. The shifts exhibit alinear dependence on denaturant concentration and we report here correctionfactors for the calculation of ‘random coil’ 1H chemicalshifts at any arbitrary denaturant concentration. Studies of arepresentative subset of peptides indicate that 13C and15N chemical shifts are also perturbed by the denaturant.These results should facilitate the application of chemical shift-basedanalytical techniques to the study of polypeptides in solution with GuHCl.The effects of the denaturant on the quality of NMR spectra and on chemicalshift referencing are also addressed.
Similar content being viewed by others
References
Arcus, V.L., Vuilleumier, S., Freund, S.M.V., Bycroft, M. and Fersht, A.R. (1995) J. Mol. Biol., 254, 305–321.
Asakura, T., Taoka, K., Demura, M. and Williamson, M.P. (1995) J. Biomol. NMR, 5, 227–236.
Bax, A. and Davis, D.G. (1985) J. Magn. Reson., 63, 207–213.
Braun, D., Wider, G. and Wüthrich, K. (1994) J. Am. Chem. Soc., {vn116}, 8466–8469.
Bruhwiler, D. and Wagner, G. (1986) J. Magn. Reson., 69, 546–551.
Bundi, A., Grathwohl, C., Hochmamn, J., Keller, R.M., Wagner, G. and Wüthrich, K. (1975) J. Magn. Reson., 18, 191–198.
Bundi, A. and Wüthrich, K. (1979) Biopolymers, 18, 285–297.
Dobson, C.M. (1992) Curr. Opin. Struct. Biol., 2, 6–12.
Englander, S.W., Sosnick, T.R., Englander, J.J. and Mayne, L. (1996) Curr. Opin. Struct. Biol., 6, 18–23.
Fiebig, K.M., Schwalbe, H., Buck, M., Smith, L.J. and Dobson, C.M. (1996) J. Chem. Phys., 100, 2661–2666.
Hoeltzli, S.D. and Frieden, C. (1995) Proc. Natl. Acad. Sci. USA, 92, 9318–9322.
Jiménez, M.A., Nieto, J.L., Rico, M., Santoro, J., Herranz, J. and Bermejo, F.J. (1986) J. Mol. Struct., 143, 435–438.
Jiménez, M.A., Blanco, F.J., Rico, M., Herranz, J., Santoro, J. and Nieto, J.L. (1992) Eur. J. Biochem., 207, 39–49.
Kay, L.E. and Bax, A. (1990) J. Magn. Reson., 86, 110–126.
Kay, L.E., Keifer, P. and Saarinen, T. (1992) J. Am. Chem. Soc., 114, 10663–10665.
Kay, M.S. and Baldwin, R.L. (1996) Nat. Struct. Biol., 3, 439–445.
Keim, P., Vigna, R.A., Marshall, R.C. and Gurd, F.R.N. (1973) J. Biol. Chem., 248, 6104–6113.
Keim, P., Vigna, R.A., Morrow, A.M., Marshall, R.C. and Gurd, F.R.N. (1974) J. Biol. Chem., 249, 4149–4156.
Kiefhaber, T., Labhardt, A.M. and Baldwin, R.L. (1995) Nature, 375, 513–515.
Kotik, M., Radford, S.E. and Dobson, C.M. (1995) Biochemistry, 34, 1714–1724.
Lu, H., Ph.D. Thesis, University of Oxford, Oxford, U.K., 1997.
Masson, A. and Wüthrich, K. (1973) FEBS Lett., 31, 114–118.
Merutka, G., Dyson, H.J. and Wright, P.E. (1995) J. Biomol. NMR, {vn5}, 14–24.
Nozaki, Y. (1972) Methods Enzymol., 26, 43–51.
Oldfield, E. (1995) J. Biomol. NMR, 5, 217–225.
Ösapay, K. and Case, D.A. (1994) J. Biomol. NMR, 4, 215–230.
Pace, C.N. (1986) Methods Enzymol., 131, 267–280.
Pastore, A. and Saudek, V. (1990) J. Magn. Reson., 90, 165–176.
Plaxco, K.W. and Dobson, C.M. (1996) Curr. Opin. Struct. Biol., 6, 630–636.
Pople, J.A. (1957a) Proc. R. Soc., 239A, 541–549.
Pople, J.A. (1957b) Proc. R. Soc., 239A, 550–556.
Pople, J.A., Schneider, W.G. and Bernstein, H.J. (1959) High-resolution Nuclear Magnetic Resonance, McGraw-Hill, New York, NY, U.S.A., pp. 400–421.
Ptitsyn, O.B. (1995) Curr. Opin. Struct. Biol., 5, 74–78.
Rance, M., Sørensen, O.W., Bodenhausen, G., Wagner, G., Ernst, R.R. and Wüthrich, K. (1983) Biochem. Biophys. Res. Commun., {vn117}, 479–485.
Redfield, C., Smith, R.A.G. and Dobson, C.M. (1994) Nat. Struct. Biol., 1, 23–29.
Reily, M.D., Thanabal, V. and Omecinsky, D.O. (1992) J. Am. Chem. Soc., 114, 6251–6252.
Richarz, R. and Wüthrich, K. (1978) Biopolymers, 17, 2133–2141.
Rizo, J., Blanco, F.J., Kobe, B., Bruch, M.D. and Gierash, L.M. (1993) Biochemistry, 32, 4881–4894.
Schulman, B.A., Redfield, C., Peng, Z.Y., Dobson, C.M. and Kim, P.S. (1995) J. Mol. Biol., 253, 651–657.
Serrano, L. (1995) J. Mol. Biol., 254, 322–333.
Shaka, A.J., Barker, P.B. and Freeman, R. (1985) J. Magn. Reson., {vn64}, 547–552.
Shin, H.-C., Merutka, G., Waltho, J.P., Wright, P.E. and Dyson, H.J. (1993) Biochemistry, 32, 6348–6355.
Shortle, D. (1993) Curr. Opin. Struct. Biol., 3, 66–74.
Shortle, D.R. (1996) Curr. Opin. Struct. Biol., 6, 24–30.
Smith, L.J., Sutcliffe, M.J., Redfield, C. and Dobson, C.M. (1991) Biochemistry, 30, 986–996.
Smith, L.J., Bolin, K.A., Schwalbe, H., MacArthur, M.W., Thornton, J.M. and Dobson, C.M. (1996) J. Mol. Biol., 225, 494–506.
Tanford, C. (1968) Adv. Protein Chem., 23, 121–282.
Thanabal, V., Omecinsky, D.O., Reily, M.D. and Cody, W.L. (1994) J. Biomol. NMR, 4, 47–59.
Wang, Y. and Shortle, D. (1995) Biochemistry, 34, 15895–15905.
Wishart, D.S., Sykes, B.D. and Richards, F.M. (1991a) FEBS Lett., {vn193}, 72–80.
Wishart, D.S., Sykes, B.D. and Richards, F.M. (1991b) J. Mol. Biol., {vn222}, 311–333.
Wishart, D.S., Sykes, B.D. and Richards, F.M. (1992) Biochemistry, {vn311}, 1647–1651.
Wishart, D.S. and Sykes, B.D. (1994a) J. Biomol. NMR, 4, 171–180.
Wishart, D.S. and Sykes, B.D. (1994b) Methods Enzymol., 239, 363–392.
Wishart, D.S., Bigam, C.G., Holm, A., Hodges, R.S. and Sykes, B.D. (1995a) J. Biomol. NMR, 5, 67–81.
Wishart, D.S., Bigam, C.G., Yao, J., Abildgaard, F., Dyson, H.J., Oldfield, E., Markley, J.L. and Sykes, B.D. (1995b) J. Biomol. NMR, 6, 135–140.
Wüthrich, K. (1986) NMR of Proteins and Nucleic Acids, Wiley, New York, NY, U.S.A., pp. 15–17.
Wüthrich, K. (1994) Curr. Opin. Struct. Biol., 4, 93–99.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Plaxco, K.W., Morton, C.J., Grimshaw, S.B. et al. The effects of guanidine hydrochloride on the ’random coil‘ conformations and NMR chemical shifts of the peptide series GGXGG. J Biomol NMR 10, 221–230 (1997). https://doi.org/10.1023/A:1018340217891
Issue Date:
DOI: https://doi.org/10.1023/A:1018340217891