Abstract
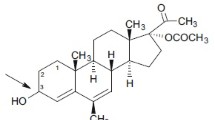
Antiprogestin activity of 5H-progesterone metabolites and their analogues 16α,17α-cyclohexan-5H-pregnan-3,20-diones was tested on rats. Modified pregnancy interruption test showed that 5α (H)-isomers of natural and synthetic hormones exhibit maximum activity.
Similar content being viewed by others
REFERENCES
A. V. Kamernitskii and I. S. Levina, Khim.-Farm. Zhurn., 25, No. 10, 4-16 (1991).
N. V. Kirpichnikova, Molecular pharmacology of antiprogestins. Abstract of Cand. Med. Sci. Dissertation, Moscow (2000).
I. S. Levina and A. V. Kamernitskii, Khim.-Farm. Zhurn., 24, No. 10, 31-39 (1999).
I. S. Levina, G. V. Nikitina, L. E. Kulikova, and A. V. Kamernitskii, Izv. AN, Ser. Khimiya, No. 3, 564-567 (1995).
A. N. Smirnov, E. V. Pokrovskaya, G. S. Kogteva, et al., Steroids, 65, No. 3, 163-170 (2000).
D. Philibert, M. Hardy, M. Gaillard-Moguilewsky, et al., J. Steroid Biochem., 34, No. 1-6, 413-417 (1989).
A. Ulmann, J. Steroid Biochem., 27, 1009-1012 (1987).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kareva, E.N., Kamernitskii, A.V., Levina, I.S. et al. Antiprogestagen Activity of 5H-Progesterone Metabolites and Their Analogues, 16α,17α-Cyclohexane-5H-Pregnan-3,20-Diones. Bulletin of Experimental Biology and Medicine 131, 340–341 (2001). https://doi.org/10.1023/A:1017995917908
Issue Date:
DOI: https://doi.org/10.1023/A:1017995917908