Abstract
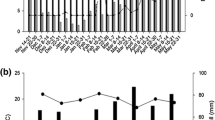
The multiplication of Soil-borne wheat mosaic virus (SBWMV) was studied in mixtures of two winter wheat (Triticum aestivum) cultivars, one susceptible (Soissons) and the other resistant (Trémie). Two seed mixtures of susceptible and resistant varieties in ratios of 1 : 1 and 1 : 3 and their component pure stands, i.e. each variety grown separately, were grown in a field infected with SBWMV. The presence of the virus was detected using DAS-ELISA from January to May. The resistant cultivar Trémie showed no foliar symptoms nor could the virus be detected in the leaves or roots. In May, about 88% of plants of susceptible cultivar Soissons grown in pure stands were infected. At this time, the disease reduction relative to pure stands was 32.2% in the 1 : 1 mixture and 39.8% in the 1 : 3 mixture. Optical density (OD) values from ELISA of the infected plants in the two mixtures were consistently lower than that of the infected plants in cultivar Soissons in pure stands. The ELISA index (EI) calculated using three scales of OD values was 65.5% in the susceptible cultivar in pure stands. The value for this index was 19.1% in the 1 : 1 mixture and 7.9% in the 1 : 3 mixture. The plants of the resistant cultivar Trémie infected in the same field and transferred in January to a growth cabinet at 15 °C multiplied the virus and produced viruliferous zoospores. These results show that the resistant cultivar Trémie plays a role in disease reduction in the cultivar mixtures in field conditions. Possible reasons for this are discussed.
Similar content being viewed by others
References
Armitage CR, Hunger RM, Sherwood JL and Weeks DL (1990) Relationship between development of hard red winter wheat and expression of resistance to wheat soilborne mosaic virus. Plant Disease 74: 356-359
Browning JAand Frey KJ (1969) Multiline cultivars as a means of disease control. Annual Review of Phytopathology 7: 355-382
Burdon JJ and Chilvers GA (1982) Host density as a factor in disease ecology. Annual Review of Plant Pathology 20: 143-166
Calonnec A, Goyeau H and de Vallavieille-Pope C (1996) Effects of induced resistance on infection efficiency and sporulation of Puccinia striiformis on seedlings in varietal mixtures and on field epidemics in pure stands. European Journal of Plant Pathology 102: 733-741
Canova A (1966) Richerche sulle malattie da virus delle Graminacee III.Polymyxa graminis Led.Vettore del virus del mosaico del Frumento. Phytopathologia Mediterranea 5: 53-58
Clark MF and Adams AN(1977) Characteristics of the microplate method of enzyme-linked immunosorbent assay for the detection of plant viruses. Journal of General Virology 34: 475-483
Clover GRG, Wright DM and Henry CM (1999) First report of soilborne wheat mosaic virus in the United Kingdom. Plant Disease 83: 880
Diao A, Chen J, Gitton F, Antoniw JF, Mullins J, Hall AM and Adams MJ (1999) Sequences of European wheat mosaic virus and Oat golden stripe virus and genome analysis of the genus furovirus. Virology 261: 331-339
Dubey SN, Brown CM and Hooker AL (1970) Inheritance of field reaction to soilborne wheat mosaic virus. Crop Science 10: 93-85
Estes AP and Brakke MK (1966) Correlation of Polymyxa graminis with transmission of soilborne wheat mosaic virus. Virology 28: 772-774
Finckh MR and Mundt CC (1992) Plant competition and disease in genetically diverse wheat populations. Oecologia 91: 82-92
Finckh MR, Gacek ES, Czembor HJ and Wolfe MS (1999) Host frequency and density effects on powdery mildew and yield in mixtures of barley cultivars. Plant Pathology 48: 807-816
Garrett KA and Mundt CC (2000) Effects of planting density and the composition of wheat cultivar mixtures on stripe rust: an analysis taking into account limits to the replication of controls. Phytopathology 90: 1313-1321
Hariri D, Courtillot M, Zaoui P and Lapierre H (1987) Multiplication of soilborne wheat mosaic virus (SBWMV) in wheat roots infected by a soil carrying SBWMV and wheat yellow mosaic virus (WYMV). Agronomie 7: 789-796
Himmel PT, Hewings AD and Glawe DA (1991) Incidence of soilborne wheat mosaic virus and its reported vector, Polymyxa graminis, in field-grown soft red winter wheat. Plant Disease 75: 1008-1012
Koenig R, Pleij CWA and Huth W (1999) Molecular characterization of a new furovirus mainly infecting rye. Archives of Virology 144: 2125-2140
Koenig R and Huth W (2000) Soil-borne rye mosaic and European wheat mosaic virus: two names for a furovirus with variable genome properties which is widely distributed in several cereal crops in Europe. Archives of Virology 145: 689-697
Lannou C, de Vallavieille-Pope C and Goyeau H (1995) Induced resistance in host mixtures and its effect on disease control in computer-simulated epidemics. Plant Pathology 44: 478-489
Lapierre H, Courtillot M, Kusiak C and Hariri D (1985) Resistance au champ des blés en semis d'automne au virus de la mosaïque du blé (wheat soilborne mosaic virus). Agronomie 5: 565-572
Merkle OG and Smith EL (1983) Inheritance of resistance to soilborne mosaic in wheat. Crop Science 23: 1075-1076
Modawi RS, Heyne EG, Brunetta P and Wilis WG (1982) Genetic studies of field reaction to wheat soilborne mosaic virus. Plant Disease 66: 1183-1184
Myers L Drumm, Sherwood JL, Siegerist WC and Hunger RM (1993) Temperature-influenced virus movement in expression of resistance to soilborne wheat mosaic virus in hard red winter wheat (Triticum aestivum). Phytopathology 83: 548-551
Nakagawa M, Soga Y, Watanable S, Gocho H and Nishio K (1959) Genetical studies of the wheat mosaic virus. II. Genes affecting the inheritance of susceptibility to strains of yellow mosaic in varietal crosses of wheat. Japanese Journal of Breeding 9: 118-120
Ohto Y and Naito S (1997) Propagation of wheat yellow mosaic virus in winter wheat under low temperature conditions. Annals of the Phytopathological Society of Japan 63: 361-365
Rao AS and Brakke MK (1969) Relation of soil-borne wheat mosaic virus and its fungal vector, Polymyxa graminis. Phytopathology 59: 581-587
Shaalan MI, Heyne EG and Sill JR (1966) Breeding wheat for resistance to soilborne wheat mosaic virus, wheat streak virus, leaf rust, stem rust and bunt. Phytopathology 56: 664-668
Shirako Y, Ali I and Wilson TMA (1990) Nucleotide sequence of soilborne wheat mosaic virus RNA II. Phytopathology 80: 1018
Trenbath BR (1977) Interactions among diverse hosts and diverse parasites. Annals of the New York Academy of Sciences 287: 124-150
van Asch MAJ, Rijkenberg FHJ and Coutinho TA (1992) Resistance induced in wheat by an avirulent race of Puccinia recondita f. sp. Tritici. Plant Disease 76: 412-415
Zhu Y, Chen H, Fan J, Wang Y, Li Y, Chen J, Fan J, Yang S, Hu L, Leung H, Mew TW, Teng PS, Wang Z and Mundt CC (2000) Genetic diversity and disease control in rice. Nature 406: 718-722
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Hariri, D., Fouchard, M. & Prud'homme, H. Incidence of Soil-borne Wheat Mosaic Virus in Mixtures of Susceptible and Resistant Wheat Cultivars. European Journal of Plant Pathology 107, 625–631 (2001). https://doi.org/10.1023/A:1017980809756
Issue Date:
DOI: https://doi.org/10.1023/A:1017980809756