Abstract
The reactions of 3-X-6-phenyl-1,2,4-triazines (X = SMe, SPh, SO2Ph) with phenylacetonitrile anion in DMF were studied. In these reactions the ring transformation product 3-amino-4,6-diphenylpyridazine, the covalent addition product 3-X-5-(α-cyanobenzyl)-6-phenyl-2,5-dihydro-1,2,4-triazine, and the ipso-substitution product 3-(1-cyano-1-phenylmethyl)-6-phenyl-1,2,4-triazine were obtained. Analogous reactions carried out in DMA gave only the addition products in excellent yields as diastereomeric mixtures of the corresponding 2,5-dihydro-1,2,4-triazines.
Similar content being viewed by others
REFERENCES
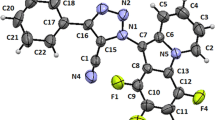
Z. Karczmarzyk, D. Branowska, A. Rykowski, and A. Fruzinski, J. Chem. Cryst. (submitted).
A. Rykowski,E. Wolinska, andH. C. Van der Plas, J. Heterocycl. Chem., 37, 879 (2000).
I. Lalezari andH. Golgolab, J. Heterocycl. Chem., 8, 689 (1970).
J. Nagy,J. Nyitrai,I. Vago, andG. I. Csonka, J. Org. Chem., 63, 5824 (1998).
M. Makosza,M. Jagusztyn-Grochowska,M. Ludwikow, andM. Jawdosiuk, Tetrahedron, 30, 3723 (1974).
A. Rykowski,O. N. Chupakhin,D. N. Kozhevnikov,V. N. Kozhevnikov,V. L. Rusinov, andH. C. van der Plas, Heterocycles, 55, 127 (2001).
V. N. Charushin,S. G. Alexeev,O. N. Chupakhin, andH. C. van der Plas, Adv. Heterocycl. Chem., 46, 73 (1989).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Rykowski, A., Wolinska, E. & van der Plas, H.C. 1,2,4-Triazine in Organic Synthesis. 16. Reactivity of 3-Substituted 6-Phenyl-1,2,4-triazines towards Phenylacetonitrile Anion in Polar Aprotic Solvents. Chemistry of Heterocyclic Compounds 37, 1418–1423 (2001). https://doi.org/10.1023/A:1017959403370
Issue Date:
DOI: https://doi.org/10.1023/A:1017959403370