Abstract
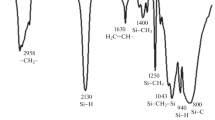
A commercial polycarbosilane (PCS) preceramic polymer has been characterised as-received and following curing under a variety of conditions. Elemental analysis, gel permeation chromatography (GPC), infra-red spectroscopy (FT-IR), simultaneous thermogravimetric analysis-differential thermal analysis (TG-DTA) and solid state nuclear magnetic resonance (NMR) have been employed. A number average molar mass of 1200 was found with a broad molar mass distribution (\(\overline M _{\text{w}} \)/\(\overline M _{\text{n}} \) = 2.97). Elemental analysis gave an empirical formula of SiC2.2H5.3O0.3. IR and Solid state 29Si and 13C NMR spectra showed the presence of Si-O-Si, SiC4, SiC3H, Si-Si, Si-CH3 and Si-CH2 groups. Simultaneous TG-DTA performed under an argon flow showed that there was a weight gain which started at approximately 240 °C. DTA showed an exotherm starting at this temperature showing that there was oxidation of the polymer even in an inert atmosphere. This is perhaps due to the oxygen in the PCS and there may also be some impurities in the inert atmosphere. Evidently the PCS is very sensitive to oxygen. Above 500 °C, weight loss dominated although the exotherm continued to approximately 700 °C. The effect of heating rate and dwell time at 200 °C on the changes in the chemical composition during curing have been explored using IR and solid state NMR spectroscopies, and elemental analysis. The longer the cure time the higher was the weight gain and greater was the extent of the oxidation reactions. Elemental analysis showed that the ratio of H and C to Si decreased with holding time at the cure-temperature while the amount of oxygen increased. Use of a higher heating rate resulted in a lower weight gain when the same holding time was used. From this it is clear that curing starts below the holding temperature.
Similar content being viewed by others
References
R. W. Rice, Ceram.Bull. 62(8) (1983) 889.
R. Riedel and W. Dressler, Ceram.Int. 22 (1996) 233.
S. Yajima, J. Hayashi and M. Omori, Chem.Lett. 9 (1975) 931.
S. Yajima, K. Okamura and J. Hayashi, ibid. 1209.
S. Yajima, C. H. Liaw, M. Omori and J. Hayashi, ibid. (1976) 435.
S. Yajima, H. Kayano, K. Okamura, M. Omori, J. Hayashi, T. Matsuzawa and K. Akutsu, Ceram. Bull. 55(12) (1976) 1065.
S. Yajima, J. Hayashi, M. Omori and K. Okamura, Nature 261 (1976) 683.
S. Yajima, K. Okamura, J. Hayashi and M. Omori, J.Amer.Ceram.Soc. 59(7/8) (1976) 324.
S. Yajima, in “Handbook of Composites,” edited by W. Watt and B. V. Perov (Elsevier science, Amsterdam, 1985) pp. 201–237.
Idem., US Patent: 4052430 (1977).
Idem., US Patent: 4100233 (1978).
G. Pouskouleli, Ceram.Int. 15 (1989) 213.
D. R. Petrak, “Polymer-Derived Ceramics,” in Engineered Materials Handbook, Vol. 4, Ceramics and Glasses, 1991, pp. 223–226.
R. Riedel, J.Eur.Ceram.Soc. 15 (1995) 703.
B. I. Lee and L. L. Hench, Mat.Res.Soc.Symp.Proc. 73 (1986) 815.
H. J. Wu and L. V. Interrante, Polym.Prepr. 33(2) (1989) 210.
T. J. Fitzgerald and A. Mortensen, J.Mater.Sci. 30(9) (1995) 1025.
M. Takeda, Y. Imai, H. Ichikawa and T. Ishikawa, Ceram.Eng.Sci.Proc. 12(7/8) (1991) 1007.
R. A. Sinclair, in “Ultrastructure Processing of Ceramics, Glasses and Composites,” edited by L. L. Hench and D. R. Ulrich (Wiley Interscience, New York, 1984) p. 256.
T. Taki, S. Maeda, K. Okamura, M. Sato and T. Matsuzawa, J.Mater.Sci.Lett. 6 (1987) 826.
T. Taki, K. Okamura and M. Sato, J.Mater.Sci. 24 (1989) 1263.
H. Suwardie, K. M. Kalyon and S. Kovenklioglu, J.Appl.Polym.Sci. 42 (1991) 1087.
H. Q. Ly, R. Taylor, R. J. Day and F. Heatley, to be published.
F. Heatley, in “NMR Spectroscopy of Polymers,” edited by R. N. Ibbett (Blackie, 1993).
K. Okamura, private communications.
E. Bouillon, F. Langlais, R. Pailler, R. Naslain, F. Cruege, P. V. Huong, J. C. Sarthou, A. Delpuech, C. Laffon, P. Lagarde, M. Monthioux and A. Oberlin, J.Mater.Sci. 26 (1991) 1333.
M. Narisawa, Bull.Chem.Soc.Jpn. 68 (1995) 1098.
Y. Hasegawa and K. Okamura, J.Mater.Sci. 21 (1986) 321.
R. M. Silverstein, G. Clayton Bassler and T. C. Morrill, in “Spectrometric Identification of Organic Compounds” (5th edition, Wiley, New York, 1991).
F. Heatley, (Department of Chemistry, University of Manchester), private communication.
(Physics and Chemistry Handbook).
Y. Hasegawa, J.Mater.Sci. 24 (1989) 1177.
G. D. Soraru, F. Babonneau and J. D. Mackenzie, ibid. 25 (1990) 3886.
K. Suzuya, K. Shibata, K. Okamura and K. Suzuki, J.Non-Cryst.Solids 150 (1992) 255.
Y. Hasegawa and K. Okamura, J.Mater.Sci. 18 (1983) 3633.
G. Chollon, M. Czerniak, R. Pailler, X. Bourrat, R. Naslain, J. P. Pilllot and R. Cannet, ibid. 32 (1997) 893.
Y. Hasegawa, M. Iimura and S. Yajima, ibid. 15 (1980) 720.
H. Ishikawa, F. Machino, S. Mitsuno, T. Ishikawa, K. Okamura and Y. Hasegawa, ibid. 21 (1986) 4352.
C. Y. Yang, P. S. Marchetti and L. V. Interrante, Polym.Prepr. 33(2) (1992) 208.
G. R. Hatfield and K. R. Carduner, J.Mater.Sci. 24 (1989) 4209.
K. Okamura, Composites 18(2) (1987) 107.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Ly, H.Q., Taylor, R., Day, R.J. et al. Conversion of polycarbosilane (PCS) to SiC-based ceramic Part 1. Characterisation of PCS and curing products. Journal of Materials Science 36, 4037–4043 (2001). https://doi.org/10.1023/A:1017942826657
Issue Date:
DOI: https://doi.org/10.1023/A:1017942826657