Abstract
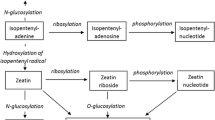
Zeatin allylic phosphate (ZAP) retarded chlorophyll loss in the barleyleaf senescence assay at a concentration 20 times higher than for6-benzyladenine (BA): the effective concentrations for ZAP and BA were 10 and 0.5 , respectively. Sodium molybdate,an inhibitor of phosphatases, decreased the ZAP effective concentration to 0.5 without affecting leaf senescence andtrans-zeatin activity in the control. This demonstrates theimportance of the phosphate group for ZAP activity or its penetration into leafcells. ZAP up-regulated the protein kinase activity of the barley leaf chromatinwith concentration dependence similar to that oftrans-zeatin. Conversely, ZAP was 1000 times less activethan trans-zeatin in the competition with anti-idiotypeantibodies (raised against antibody to zeatin) for binding with atrans-zeatin-binding site oftrans-zeatin-binding protein ZBP67 isolated from barleyleaves. In contrast to trans-zeatin, ZAP did not activateRNA synthesis in the presence of ZBP in the in vitro systemcontaining chromatin and RNA polymerase I isolated from barley leaves. Insummary, data presented show that ZAP possesses cytokinin activity asdemonstrated by the retardation of barley leaf senescence, but moleculartarget(s) for ZAP in barley leaf cells differs, at least partially, from thesefor trans-zeatin. It seems possible that the cytokininactivity of ZAP results from its hydrolysis while producing zeatin.
Similar content being viewed by others
References
Auer C.A. 1997. Cytokinin conjugation: recent advances and patterns in plant evolution. Plant Growth. Regul. 23: 17–32.
Bassil N.V., Mok D.W.S. and Mok M.C. 1993. Partial purification of a cis-trans-isomerase of zeatin from immature seeds of Phaseolus vulgaris L. Plant Physiol. 102: 867–872.
Hare P.D. and van Staden J. 1997. The molecular basis of cytokinin action. Plant Growth. Regul. 23: 41–78.
Jones R.J. and Schreiber B.M.N. 1997. Role and function of cytokinin oxidase in plants. Plant Growth. Regul. 23: 123–134.
Kakimoto T. 1996. CKI1, a histidine kinase homolog implicated in cytokinin signal transduction. Science. 274: 982–985.
Karavaiko N.N., Zemlyachenko Ya.V., Selivankina S.Yu. and Kulaeva O.N. 1995.Isolation of cytokinin-binding protein involved in the activation of in vitro RNA synthesis by transzeatin from barley cytosol. Russian J. Plant Physiol. 42: 481–487.
Kulaeva O.N. 1973. Cytokinins, their Structure and Functions. Nauka, Moscow.
Kulaeva O.N., Karavaiko N.N., Moshkov I.E., Selivankina S.Yu. and Novikova G.V. 1990. Isolation of a protein with cytokininreceptor properties by means of anti-idiotype antibodies. FEBS Lett. 261: 410–412.
Kulaeva O.N., Karavaiko N.N., Selivankina S.Yu., Moshkov I.E., Novikova G.V., Zemlyachenko Ya.V. et al. 1996. Cytokinin signalling systems. Plant Growth. Regul. 18: 29–37.
Kulaeva O.N., Karavaiko N.N., Selivankina S.Yu., Zemlyachenko Ya.V. and Shipilova C.V. 1995. Receptor of trans-zeatin involved in transcription activation by cytokinin. FEBS. Lett. 366: 26–28.
Kulaeva O.N., Zagranichnaya T.K., Brovko F.A., Karavaiko N.N., Selivankina S.Yu., Zemlyachenko Ya.V. et al. 1998. A new family of cytokinin receptor from cereals. FEBS. Lett. 423: 239–242.
Letham D.S. and Palni L.M.S. 1983. The biosynthesis and metabolism of cytokinins. Annu. Rev. Plant Physiol. 34: 163–197.
Marx J. 1985. Making antibodies without the antigenes. Science. 228: 162–165.
Miller C.O. 1961. Kinetin and related compounds in plant growth. Annu. Rev. Plant Physiol. 12: 395–408.
Plakidou-Dymock S., Dymock D. and Hooley R. 1998. A higher plant seven transmembrane receptor homologue that influences a sensitivity to cytokinins. Curr. Biol. 8: 315–324.
Prinsen E., Kaminek M. and van Onckelen H.A. 1997. Cytokinin biosynthesis: a black box?. Plant Growth. Regul. 23:3–15.
Selivankina S.Yu., Romanko E.G., Novikova G.V., Muromtseva D.G. and Kulaeva O.N. 1998. Effect of cytokinin and other phytohormones on protein kinases associated with chromatin and RNA polymerase I in barley leaves. Soviet Plant Physiol. 35: 205–212.
Selivankina S.Yu., Romanko E.G., Ovcharov A.K. and Kharchenko V.I. 1982. Activation of RNA synthesis in vitro by cytokininreceptor complex from barley leaves. Soviet Plant Physiol. 26: 1016–1028.
Shaw G., Smallwood B.M. and Wilson D.V. 1967. Phosphorylated derivatives of the cytokinins zeatin and its 9-L-ribofuranoside: Naturally occurring adenine and adenosine derivatives with plant cell division promoting activity. Experimentia. 23: 515–518.
Skoog F. and Miller C.O. 1957. Chemical regulation of growth and organ formation in plant tissue cultured in vitro. Sympos. Soc. Exptl. Biol. 11: 118–231.
Vonk C.R. and Davelaar E. 1992. Zeatin allylic phosphate: Occurrence, formation and possible role. In: Karssen C.M., van Loon L.C. and Vreugdenhil D. (eds), Progress in Plant Growth Regulation. Kluwer Academic Publishers, Dordrecht, pp. 617–623.
Vonk C.R., Davelaar E. and Ribot S.A. 1989. Formation of zeatin allylic phosphate by the microsomal fraction of bulb disks of Iris x hollandica Tub. and tubers of Heliantus tuberosus L. Plant Growth. Regul. 8: 263–276.
Zemlyachenko Ya.V., Karavaiko N.N., Maslova G.G. and Kulaeva O.N. 1997. A new approach for studying the structure-function relationships in cytokinins. Doklady Academii. Nauk. 353: 261–263.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Selivankina, S., Karavaiko, N., Kuiper, D. et al. Cytokinin activity of zeatin allylic phosphate, a natural compound. Plant Growth Regulation 33, 157–164 (2001). https://doi.org/10.1023/A:1017553023145
Issue Date:
DOI: https://doi.org/10.1023/A:1017553023145