Abstract
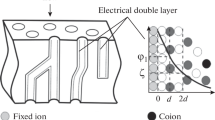
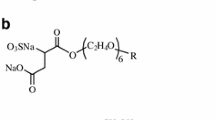
The effect of organic surfactants (1,4-butynediol, benzoic and disulfonaphthalene acids) on electrochemical properties of heterogeneous ion-exchange membranes MK-40 and MA-40 is studied. The surfactants effect the membrane properties both under equilibrium conditions and, especially, in the electric field, suggesting that they are involved in both the exchange sorption and the electrotransport. The mechanism of their effect on the anionite membrane is considered. It is shown that bipolar boundaries can arise in the membrane, promoting water dissociation and transport of hydrogen and hydroxyl ions in the field direction.
Similar content being viewed by others
REFERENCES
Shishkina, S.V., Maslennikova, I.Yu., and Alalykina, I., Elektrokhimiya, 1996, vol. 32, p. 290.
Gnusin, N.P., Berezina, N.P., and Kononenko, N.A., Elektrokhimiya, 1987, vol. 23, p. 142.
Berezina, N.P., Demina, O.A., and Timofeev, S.V., Elektrokhimiya, 1992, vol. 28, p. 463.
Berezina, N.P., Kononenko, N.A., and Kukora, T.V., Izv. Vyssh. Uchebn. Zaved., Khim. Khim. Tekhnol., 1987, vol. 30, p. 75.
Berezina, N.P., Stenina, E.V., and Fedorovich, N.V., Elektrokhimiya, 1987, vol. 23, p. 811.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Shishkina, S.V., Kovyazina, L.I., Maslenikova, I.Y. et al. Interaction of Organic Surfactants with Heterogeneous Ion-Exchange Membranes. Russian Journal of Electrochemistry 38, 892–895 (2002). https://doi.org/10.1023/A:1016822030399
Issue Date:
DOI: https://doi.org/10.1023/A:1016822030399