Abstract
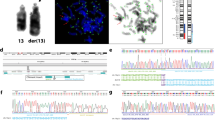
Robertsonian translocations (ROBs) are the most common rearrangements in humans, contributing significantly to genetic imbalance, fetal wastage, mental retardation and birth defects. Rob(14q21q) and rob(13q14q), which are formed predominantly during female meiosis, comprise the majority (∼85%) of all ROBs. Previous studies have shown that the breakpoints are consistently located within specific regions of the proximal short arms of chromosomes 13, 14, and 21. The high prevalence of these translocations, the consistent breakpoints found, and the fact that roughly 50% of cases occur sporadically suggest that the sequences at or near the breakpoints confer susceptibility to chromosome rearrangement and that the rearrangements occur through a specific mechanism. To investigate this hypothesis, we developed hamster–human somatic cell hybrids derived from de novo rob(14q21q) patients that contained the translocated chromosome segregated from the other acrocentric chromosomes. We determined the physical order of five satellite III subfamilies on 14p, and investigated their involvement in formation of these de novo translocations.
Similar content being viewed by others
References
Arnheim N (1983) Concerted evolution of multigene families. In: Nei M, Koehn RK, eds. Evolution of Genes and Proteins. Sunderland: Sinauer Associates Inc, pp 3–61.
Arnheim N, Krystal M, Schmickel R, Wilson G, Ryder O, Zimmer E (1980) Molecular evidence for genetic exchanges among ribosomal genes on nonhomologous chromosomes in man and apes. Proc Natl Acad Sci USA 77: 732–7327.
Bandyopadhyay R, McQuillan C, Page SL, Choo KHA, Shaffer LG (2001) Identi¢cation and characterization of sat-ellite III subfamilies to the acrocentric chromosomes. Chromosome Res 9: 22–233.
Choo KH, Vissel B, Brown R, Filby RG, Earle E (1988) Hom-ologous alpha satellite sequences on human acrocentric chromosomes with selectivity for chromosomes 13, 14 and 21: implications for recombination between nonhomologues and Robertsonian translocations. Nucleic Acids Res 16: 127–1284.
Choo KH, Earle E, McQuillan C (1990) A homologous sub-family of satellite III DNA on human chromosomes 14 and 22. Nucleic Acids Res 18: 564–5648.
Choo KH, Earle E, Vissel B, Kalitsis P (1992) A chromosome 14–speci¢c human satellite III DNA subfamily that shows variable presence on different chromosomes 14. Am J Hum Genet 50: 70–716.
Collins FS, Patrinos A, Jordan E, Chakravarti A, Gesteland R, Walters L (1998) New goals for the U.S. Human Genome Project: 199–2003. Science 282: 68–689.
Dale S, Earle E, Voullaire L, Rogers J, Choo KH (1989) Centromeric alpha satellite DNA ampli¢cation and translo-cation in an unusually large chromosome 14p+ variant. Hum Genet 82: 15–158.
Earle E, Dale S, Choo KH (1989) Ampli¢cation of satellite III DNA in an unusually large chromosome 14p+ variant. Hum Genet 82: 18–190.
Earle E, Shaffer LG, Kalitsis P, McQuillan C, Dale S, Choo KH (1992) Identi¢cation of DNA sequences £anking the breakpoint of human t(14q21q) Robertsonian translocations. Am J Hum Genet 50: 71–724.
Gosden JR, Mitchell AR, Buckland RA, Clayton RP, Evans HJ (1975) The location of four human satellite DNAs on human chromosomes. Exp Cell Res 92: 14–158.
Gosden JR, Lawrie SS, Gosden CM (1981) Satellite DNA sequences in the human acrocentric chromosomes: infor-mation from translocations and heteromorphisms. Am J Hum Genet 33: 24–251.
Gravholt CH, Friedrich U, Caprani M, Jorgensen AL (1992) Breakpoints in Robertsonian translocations are localized to satellite III DNA by £uorescence in-situ hybridization. Genomics 14: 92–930.
Greig GM, Willard HF (1992) Beta satellite DNA: charac-terization and localization of two subfamilies from the distal and proximal short arms of the human acrocentric chromosomes. Genomics 12: 57–580.
Hamerton JL, Canning N, Ray M, Smith S (1975) A cytogenetic survey of 14,069 newborn infants. Incidence of chromosome abnormalities. Clin Genet 8: 22–243.
Han JY, Choo KH, Shaffer LG (1994) Molecular cytogenetic characterization of 17 rob(13q14q) Robertsonian translo-cations by FISH, narrowing the region containing the breakpoints. Am J Hum Genet 55: 96–967.
Hassold TJ (1985) The origin of aneuploidy in humans. Basic Life Sci 36: 10–116.
Hassold TJ, Jacobs PA (1984) Trisomy in man. Annu Rev Genet 18: 6–97.
Hecht F, Kimberling WJ (1971) Patterns of D chromosome involvement in human (DqDq) and (DqGq) Robertsonian rearrangements. Am J Hum Genet 23: 36–367.
Jacobs PA (1981) Mutation rates of structural chromosome rearrangements in man. Am J Hum Gene 33: 4–54.
Jones KW, Corneo G (1971) Location of satellite and homo-geneous DNA sequences on human chromosomes. Nat New Biol 233: 26–271.
Kalitsis P, Earle E, Vissel B, Shaffer LG, Choo KH (1993) A chromosome 13–speci¢c human satellite I DNA subfamily Satellite III sequences on 14p 241 with minor presence on chromosome 21: further studies on Robertsonian translocations. Genomics 16: 10–112.
Miller OJ (1981) Role of the nucleolus organizer in the etiology of Down Syndrome. In: de la Cruz F, Gerald P eds. Trisomy 21 (Down Syndrome): Research Perspective. Baltimore, MD: University Park Press, pp 16–176.
Ohno S, Trujillo JM, Kaplan WD, Kinosita R (1961) Nucleolus-organizer in the causation of chromosome abnormalities. Lancet 2: 12–126.
Page SL, Shin JC, Han JY, Choo KH, Shaffer LG (1996) Breakpoint diversity illustrates distinct mechanisms for Robertsonian translocation formation. Hum Mol Genet 5: 127–1288.
Page SL, Shaffer LG (1997) Nonhomologous Robertsonian translocations form predominantly during female meiosis. Nat Genet 15: 23–232.
Rowley JD, Pergament E (1969) Possible non random selection of D group chromosomes involved in centric-fusion translocations. Ann Genet 12: 17–183.
Shaffer LG, Lupski JR (2000) Molecular mechanisms for con-stitutional chromosomal rearrangements in humans. Annu Rev Genet 34: 29–329.
Sullivan BA, Jenkins LS, Karson EM, Leanna-Cox J, Schwartz S (1996) Evidence for structural heterogeneity from molecu-lar cytogenetic analysis of dicentric Robertsonian trans-locations. Am J Hum Genet 59: 16–175.
Tagarro I, Fernandez-Peralta AM, Gonzalez-Aguilera JJ (1994a) Chromosomal localization of human satellites 2 and 3 by a FISH method using oligonucleotides as probes. Hum Genet 93: 38–388.
Tagarro I, Wiegant J, Raap AK, Gonzalez-Aguilera JJ, Fernandez-Peralta AM (1994b) Assignment of human satel-lite 1 DNA as revealed by £uorescent in-situ hybridization with oligonucleotides. Hum Genet 93: 12–128.
Therman E (1980) Human Chromosomes. Structure, Behavior, Effects, 1st edn. New York, Heidelberg and Berlin: Springer-Verlag, pp 28–301.
Therman E, Susman B, Denniston C (1989) The non-random participation of human acrocentric chromosomes in Robertsonian translocation. Ann Hum Genet 53: 4–65.
Trowell HE, Nagy A, Vissel B, Choo KH (1993) Long-range analyses of the centromeric regions of human chromosomes 13, 14 and 21: identi¢cation of a narrow domain containing two key centromeric DNA elements. Hum Mol Genet 2: 163–1649.
Tuck-Muller CM, Bordson BL, Varela M, Bennett JW (1984) NOR associations with heterochromatin. Cytogenet Cell Genet 38: 16–170.
Wang SY, Cruts M, Del-Favero J et al. (1999) A high-resolution physical map of human chromosome 21p using yeast arti¢cial chromosomes. Genome Res 9: 105–1073.
Waye JS, Willard HF (1989) Human beta satellite DNA: genomic organization and sequence de¢nition of a class of highly repetitive tandem DNA. Proc Natl Acad Sci USA 86: 625–6254.
Zoghbi HY, Sandkuyl LA, Ott J et al. (1989) Assignment of autosomal dominant spinocerebeller ataxia (SCA1) centromeric to the HLA region on the short arm of the chromosome 6, using multilocus linkage analysis. Am J Hum Genet 44: 25–263.
Rights and permissions
About this article
Cite this article
Bandyopadhyay, R., Berend, S.A., Page, S.L. et al. Satellite III sequences on 14p and their relevance to Robertsonian translocation formation. Chromosome Res 9, 235–242 (2001). https://doi.org/10.1023/A:1016652621226
Issue Date:
DOI: https://doi.org/10.1023/A:1016652621226