Abstract
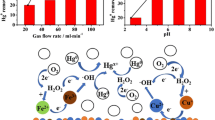
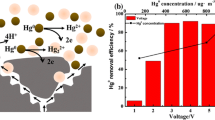
Electrochemical cells are described, which allow the oxidative removal of gaseous atomic mercury from gas streams. The gas stream is fed into an electrochemical cell containing an electrolyte solution and a three-dimensional anode with a large surface. The results can be explained by assuming that the atomic mercury is first transferred from the gas to the electrolyte solution followed by oxidation to mercury(II) ions at the electrode surface. Once mercury(II) has reached a sufficient concentration, it reacts with the atomic mercury to form mercury(I). The dissolved mercury(I) is than oxidized to mercury(II) at the electrodes. Therefore the efficiency of mercury removal from the gas stream can be enhanced by adding mercury(II) to the electrolyte at the very beginning of the process.
Similar content being viewed by others
References
G. Kreysa and H.J. Külps, Chem.-Ing.-Tech. 55 (1983) 58.
G. Kreysa and K. Jüttner, in F. Lapicque, A. Storck and A.A. Wragg (Eds), ‘Electrochemical Engineering and Energy’ (Plenum, New York, 1994), p. 255.
K. Sundmacherand U. Hoffmann, Chem. Eng. Sci. 54 (1999) 2937.
P. Neumann and G. Schmidt, Umwelt 22 (1992) 574.
R. Kubisa, R. Holste and R. Hillen, Lecture presented at IUTA Duisburg (1994).
A. Iverfeldt and O. Linquist, Atmos. Env. 20 (1986) 1567.
G. Petersen, A. Iverfeldt and J. Munthe, Atmos. Env. 29 (1995) 47.
G.Ya. Gerasimov, High Energy Chem. 34 (2000) 13.
F. Scholz and M. Lovric, Electroanal. 8 (1996) 1075.
F. Scholz and M. Lovric, Electroanal. 9 (1997) 1189.
H.K. Hansen, L.M. Ottosen, B.K. Kleim and A. Villumsen, J. Chem. Tech. Biotechnol. 70 (1997) 67.
W. Yong-Fu, C. Wie-Lung and Y. Shi-Chern, J. Environ. Sci. Health A35 (2000) 1153.
F. Scholz and B. Meyer, in A.J. Bard and I. Rubinstein (Eds), ‘Electroanalytical Chemistry, A Series of Advances’ (Marcel Dekker, New York) 20 (1998) 1.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kabbe, C., Kauschka, G. & Scholz, F. Gas purification by removal of atomic mercury using electrochemical cells. Journal of Applied Electrochemistry 32, 379–382 (2002). https://doi.org/10.1023/A:1016395328952
Issue Date:
DOI: https://doi.org/10.1023/A:1016395328952