Abstract
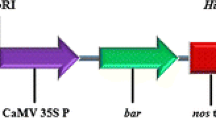
In the co-inoculation technique, genetic transformation is performed using a mixture of Agrobacterium strains – shoot regeneration is induced by the wild-type strain 82.139, while the transferable genes are provided in a binary plasmid by another, non-oncogenic Agrobacterium strain. The aim of the present work was to study the applicability of co-inoculation under both in vitro and greenhouse conditions for in planta transformation in silver birch (Betula pendula Roth). In addition to the original method, several modifications of the technique including an genetically engineered 82.139 strain harbouring the binary pGUSINT were tested. The co-inoculations resulted in a gall formation frequency of 52–94% with greenhouse seedlings, and 4–63% with tissue-cultured plantlets, the shoot induction percentage varying by 0–13 in the greenhouse and 42–75 in vitro. PCR analysis verified that the majority of the regenerated material was non-transgenic, with a few individuals showing integration of the oncogenic T-DNA. According to the histochemical tests, however, some of the numerous differentiating buds and small shoots on gall tissues were transgenic, and contained the GUS reporter gene. The results show that it would have been necessary to apply selection pressure during differentiation in order to recover shoots transformed with the desired genes from the binary plasmid. The morphology and growth of all the regenerated plantlets was normal, suggesting that the oncogenic T-DNA was not expressed even though it was present. In conclusion, it was possible to obtain transgenic silver birch plantlets using the A. tumefaciens strain 82.139, but the co-inoculation method is not directly applicable as in planta transformation protocol.
Similar content being viewed by others
References
Agrios CN (1988) Plant Pathology. Academic Press, London
Aronen T & Häggman H (1995) Differences in Agrobacterium infections in silver birch and Scots pine. Eur. J. For. Pathol. 25: 197–213
Azmi A, Dewitte W, Drevet C, van Onckelen H, Landré P, Boudet AM, Jouanin L & Chriqui D (1997) Bud regeneration from Eucalyptus globulus clones and seedlings through hormonal imbalances induced by Agrobacterium tumefaciens strain 82.139. Plant Sci. 127: 81–90
Brasileiro ACM, Leple J-C, Ounnoughi D, Michel M-F & Jouanin L (1991) An alternative approach for gene transfer in trees using wild-type Agrobacterium strains. Plant Mol. Biol. 17: 441–452
Brasileiro ACM, Tourneur C, Leple J-C, Combes V & Jouanin L (1992) Expression of the mutant Arabidopsis thaliana acetolactate synthase gene confers chlorsulfuron resistance to transgenic poplar plants. Transgenic Res. 1: 133–141
Charest PJ, Devantier Y & Lachance D (1996) Stable genetic transformation of Picea mariana (black spruce) via particle bombardment. In Vitro Cell. Dev. Biol. Plant 32: 91–99
Chee PP, Fober AK & Slightom LJ (1989) Transformation of soybean (Glycine max L.) by infecting germinating seeds with Agrobacterium tumefaciens. Plant Physiol. 91: 1212–1218
Doyle JJ & Doyle JL (1990) Isolation of plant DNA from fresh tissue. BRL Focus 12: 13–15
Drevet C, Brasileiro ACM & Jouanin L (1994) Oncogene arrangement in a shooty strain of Agrobacterium tumefaciens. Plant Mol. Biol. 25: 83–90
Deblaere R, Bytebier B, De Greve H, Deboeck F, Schell J, Van Montagu M & Leemans O (1985) Efficient octopine ti plasmid-derived vectors for Agrobacterium-mediated gene transfer to plants. Nucleic Acids Res. 13: 3777–3788
Feldman KA & Marks MD (1987) Agrobacterium-mediated transformation of germinating seeds of Arabidopsis thaliana:a non-tissue culture approach. Mol. Gen. Genet 208: 1–9.
Fenning TM, Tymens SS, Gartland JS, Brasier CM & Gartland KMA (1996) Transformation and regeneration of English elm using wild-type Agrobacterium tumefaciens. Plant Sci. 116: 37–46
Grant NJ, Fenning TM & Hammatt N (1998) Regeneration and transformation of wild cherry (Prunus avium L.) and bird cherry (Prunus padus L.). In: Davey MR, Alderson PG, Lowe KC & Power JB (eds) Tree Biotechnology: Towards the Millenium (pp 249–258). Nottingham University Press, UK
Häggman HM, Aronen TS & Nikkanen TO (1997) Gene transfer by particle bombardment to Norway spruce and Scots pine pollen. Can. J. For. Res. 27: 928–935
Jefferson RA (1987) Assaying chimeric genes in plants: the GUS gene fusion system. Plant Mol. Biol. Rep. 5: 387–405
Keinonen-Mettälä K, Pappinen A & von Weissenberg K (1998) Comparisons of the effiency of some promoters in silver birch (Betula pendula). Plant Cell Rep. 17: 356–361
Lemmetyinen J, Keinonen-Mettälä K, Lännenpää M, von Weissenberg K & Sopanen T (1998) Activity of the CaMV35S promoter in various parts of transgenic early flowering birch clones. Plant Cell Rep. 18: 243–248
MacKay J, Séquin A & Lalonde M (1988) Genetic transformation of 9 in vitro clones of Alnus and Betula by Agrobacterium tumefaciencis. Plant Cell Rep. 7: 229–232
Michel M-F, Brasileiro ACM, Depierreux C, Otten L, Delmotte F & Jouanin L (1990) Identification of different Agrobacterium strains isolated from the same forest nursery. Appl. Environ. Microbiol. 56: 3537–3545
Miller JH (1972) Experiments in Molecular Genetics. Cold Spring Harbor, New York
Mohri T, Mukai Y & K Shinohara (1997) Agrobacterium tumefaciens-mediated transformation of Japanese white birch (Betula platyphylla var. japonica). Plant Sci. 127: 53–60
Regier DA, Akiyoshi DE & Gordon MP (1989) Nucleotide sequence of the tzs gene from Agrobacterium rhizogenes strain A4. Nucleic Acids Res. 17: 8885
Rezmer C, Schlichting R, Wächter R & Ullrich CI (1999) Identification and localization of transformed cells in Agrobacterium tumefaciens-induced plant tumors. Planta 209: 399–405
Robertson D, Weissinger AK, Ackley R, Glover S & Sederoff RR (1992) Genetic transformation of Norway Spruce (Picea abies (L) Karst) using somatic embryo explants by microprojectile bombardment. Plant Mol. Biol. 19: 925–935
Rohini VK & Sankara Rao K (2000) Transformation of peanut (Arachis hypogaea L.): a non-tissue culture based approach for generating transgenic plants. Plant Sci. 150: 41–49
Stomp, AM (1992) Histochemical localization of β-glucuronidase. In: Gallagher SR (ed) GUS Protocols. Using the GUS gene as a Reporter of Gene Expression (pp 103–114). Academic Press, San Diego, California
Tepfer M & Casse-Delbart F (1987) Agrobacterium rhizogenes as a vector for transforming higher plants. Microbiol. Sci. 4: 24–28
Tervonen A (1981) Experiments in vegetative propagation by cuttings of broadleaf trees at Haapastensyrjä. The Foundation for Forest Tree Breeding in Finland, Information 3/1981. ISSN 0357–7171
Valjakka M, Aronen T, Kangasjärvi J, Vapaavuori E & Häggman H (2000) Genetic transformation of silver birch (Betula pendula) by particle bombardment. Tree Physiol. 20: 607–613
Vancanneyt G, Schmidt R, O'Connor-Sanchez A, Willmitzer L & Rocha-Sosa M (1990) Construction of an intron-containing marker gene: Splicing of the intron in transgenic plants and its use in monitoring early events in Agrobacterium-mediated plant transformation. Mol. Gen. Genet. 220: 245–250
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Aronen, T.S., Häggman, J.H. & Häggman, H.M. Applicability of the co-inoculation technique using Agrobacterium tumefaciens shooty-tumour strain 82.139 in silver birch. Plant Cell, Tissue and Organ Culture 70, 147–154 (2002). https://doi.org/10.1023/A:1016317904229
Issue Date:
DOI: https://doi.org/10.1023/A:1016317904229