Abstract
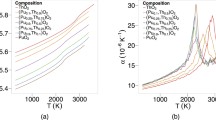
The electrical conductivity of YBa2Cu3O7 – δ is found to change sharply near the T–OII and OII–OI structural phase transitions. This finding is interpreted using Kroger's quasi-chemical reactions and a phenomenological model of oxygen nonstoichiometry which considers the formation of oxygen divacancies at T < 750 K and δ < 0.5.
Similar content being viewed by others
REFERENCES
Smith, D.M., Defects and Structural Changes in Perovskite Systems: From Insulator to Superconductors, Cryst. Lattice Defects, 1989, vol. 18, nos. 1–3, pp. 355–375.
Nakamura, K., Gufan, A.Yu., Gufan, Yu.M., et al., Effect of Structural Phase Transitions on the State of Cooper Condensate in Yba2Cu3O7 – δ, Kristallografiya, 1999, vol. 44, no. 4, pp. 650–656.
Kiselev, A.A., Vysokotemperaturnaya sverkhprovodimost' (High-Temperature Superconductivity), Leningrad: Mashinostroenie, 1990, pp. 378–379.
Lavrov, A.N. and Kozeeva, L.P., Effect of Low-Temperature Oxygen Ordering on the Properties of RbBa2Cu3O6 + x (R = Y, Tm, Lu) Single Crystals, Neorg.Mater., 1998, vol. 34, no. 11, pp. 1394–1403 [Inorg.Mater. (Engl. Transl.), vol. 34, no. 11, pp. 1177–1185].
Ryelandt, L., Schewebach, A., Willot, I., et al., Reactive Growth of YBa2Cu3O7 Ð δLayers on Y2BaCuO5 Substrates, J. Alloys Compd., 1993, vol. 20, no. 195, pp. 227–230.
Shambalev, V.N., Kalanda, N.A., Pan'kov, V.V., et al., Belarus Patent 2038430.
Shuman, P.A. and Sheiner, L.S., Precision Small-Separation Four-Point Probe, Prib. Nauchn. Issled., 1964, no. 8, pp. 25–28.
Bode, M., Teske, K., and Ullmann, H., Fest-Elektrolyt Coulonometrie (FEC). Einevielseitige Methode zur Untersuchung von Gas-Festkorper-Wechselwirkungen, Fachz. Lab., 1994, vol. 38, pp. 495–500.
Leonidov, L.A., Blonovskov, Ya.N., and Flayatau, E.E., Transport Properties and Defect Structure of YBa2Cu3O7 – δ, Physica C (Amsterdam), 1989, vol. 158, no. 1/2, pp. 287–292.
Carrillo-Cabrera, W., Wiemhofer, H.-D., and Gopel, W., Ionic Conductivity of Oxygen Ions in YBa2CuO3O7 – x , Solid State Ionics, 1989, vol. 32, no. 33, pp. 1172–1178.
Shining, Z., Xigkui, Z., Ziran, X., et al., The Linear Temperature Behavior of Resistivity and Oxygen Stoichiometry in YBa2Cu3O7 – x , Chin. Phys. Lett., 1989, vol. 6, no. 4, pp. 185–188.
Fiory, A.T., Gurvich, M., and Cava, R.J., Effect of Oxygen Desorption on Electrical Transport in YBa2Cu3O7 – δ, Phys. Rev. B: Condens. Matter, 1987, vol. 36, no. 13, pp. 7262–7265.
Novotny, J. and Rekas, M., Defect Equilibria and Transport in YBa2Cu3O7 – δat Elevated Temperatures, J. Am.Ceram. Soc., 1990, vol. 73, no. 4, pp. 1054–1061.
Kovtunenko, P.V., Fizicheskaya khimiya tverdogo tela: Kristally s defektami (Physical Chemistry of Solids: Imperfect Crystals), Moscow: Vysshaya Shkola, 1993, pp. 176–177.
Marucco, J.F. and Gledel, C., Non-Stoichiometry and Thermodynamics of Defects in the System YBa2Cu3Ox, Physica C (Amsterdam), 1989, vol. 160, pp. 73–79.
Novotny, J., Rekas, M., and Weppner, W., Defect Equilibria and Transport in YBa2Cu3O7 – δ at Elevated Temperatures, J. Am. Ceram. Soc., 1990, vol. 73, no. 4, pp1048–1053.
Verveij, H., Defect Chemistry of YBa2Cu3Ox Strong Evidence for the Presence of Cu+, Solid State Commun., 1987, vol. 164, no. 9, pp. 1213–1216.
Lee, B. and Lee, D., Thermodynamic Evolution for the Y2O3–BaO–CuOx System, J. Am. Ceram. Soc., 1991, vol. 74, no. 1, pp. 78–84.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kalanda, N.A., Trukhan, V.M. & Marenkin, S.F. Electrical Transport and Phenomenological Model of Oxygen Nonstoichiometry in YBa2Cu3O7 – δ . Inorganic Materials 38, 694–699 (2002). https://doi.org/10.1023/A:1016292409175
Issue Date:
DOI: https://doi.org/10.1023/A:1016292409175