Abstract
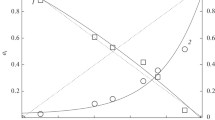
The equilibrium vapor composition in the Pb–I2 system at temperatures from 400 to 2000 K and total pressures from 102 to 105 Pa was assessed by thermodynamic analysis. The results show that the dominant vapor species is PbI2 . PbI2 dissociation is significant starting at 1000–1300 K, depending on the total pressure in the system.
Similar content being viewed by others
REFERENCES
Indutnyi, I.Z., Kostyshin, M.T., and Kasyarum, O.P., Fotostimulirovannye vzaimodeistviya v strukturakh metall–poluprovodnik (Photoinduced Reactions in Metal–Semiconductor Structures), Kiev: Naukova Dumka, 1992.
Shieber, M., James, R.B., Lund, J.C., et al., State of the Art of Wide-Band Gap Semiconductor Nuclear Radiation Detectors, Nuovo Cimento Soc. Ital. Fis., A, 1996, vol. 109, no. 9, pp. 1253–1260.
Rybak, V.M., Kalush, A.Z., and Logush, O.I., USSR Inventor's Certificate no. 1358487, Byull. Izobret., 1987, no. 45, p. 44.
Ruzinov, L.P. and Gulyanitskii, B.S., Ravnovesnye prevrashcheniya metallurgicheskikh reaktsii (Equilibrium Transformations of Metallurgical Reactions), Moscow: Metallurgiya, 1975.
Rolsten, R.F., Iodide Metals and Metal Iodides, New York: Wiley, 1961. Translated under the title Iodidnye metally i iodidy metallov, Moscow: Metallurgiya, 1968.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Rybak, O.V., Kurilo, I.V. Equilibrium Vapor Composition in the Pb–I2 System. Inorganic Materials 38, 735–737 (2002). https://doi.org/10.1023/A:1016256828740
Issue Date:
DOI: https://doi.org/10.1023/A:1016256828740