Abstract
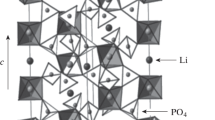
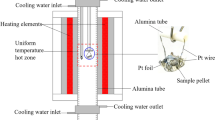
Crystallization in the LiOH–GeO2–H2O and LiOH–SiO2–H2O hydrothermal systems was studied at 500°C and 0.1 GPa. The systems were shown to contain both the isostructural compounds Li2SiO3 and Li2GeO3 and phases differing in crystal chemistry: Li2Si2O5, Li2Ge3O6(OH)2, and Li3HGe7O16 · 4H2O (containing Ge in different oxygen coordinations). The crystallization fields revealed in the germanate system are (in order of increasing LiOH concentration) GeO2 ⇒ GeO2+ Li2Ge3O6(OH)2 ⇒ Li2Ge3O6(OH)2 + Li2GeO3⇒ Li3HGe7O16 · 4H2O + Li2GeO3; those in the silicate system are α-SiO2 ⇒ α-SiO2+ Li2Si2O5 ⇒ Li2Si2O6 + Li2SiO3 ⇒ Li2SiO3. Increasing the LiOH concentration increases the number of Li atoms per tetrahedrally coordinated Si or Ge atom in the crystallizing compounds. The high stability of Li2Ge3O6(OH)2 is interpreted in terms of the matrix assembly of the structure from cyclic invariant subpolyhedral structural units.
Similar content being viewed by others
REFERENCES
Ilyushin, G.D. and Dem'yanets, L.N., Germanaty chetyrekhvalentnykh metallov (Germanates of Tetravalent Metals), Moscow: VINITI, 1989.
Ilyushin, G.D. and Dem'yanets, L.N., Crystal Chemistry of Germanates: Structures of Li,Ge-Germanates, Kristallografiya, 2000, vol. 45, no. 4, pp. 684–691.
Ilyushin, G.D. and Dem'yanets, L.N., Crystal Chemistry of Framework Li,GeVI-Germanates: Combinatorial– Topological Analysis and Structural Modeling, Kristallografiya, 2001, vol. 46, no. 5, pp. 875–883.
Nevskii, N.N., Ivanova, L.N., Ilyukhin, V.V., and Belov, N.V., Crystal Structure of a New Lithium Germanate, Li2Ge[GeO3]2(OH), Dokl. Akad. Nauk SSSR, 1978, vol. 242, no. 6, pp. 1330–1333.
Klaska, R. and Jendryan, R., Zur Struktur von Li2GeGe2O6(OH)2 und seiner Verwachssung mit LiBGeO4, Z. Kristallogr., 1983, vol. 162, nos. 1–4, pp. 129–131.
Wittmann, A. and Modern, E., Untersuchungen im Zweistoff Li2O–GeO, Monatsh. Chem., 1965, vol. 92, no. 2, pp. 581–582.
Völlenkle, H., Wittmann, A., and Nowotny, H., Die Kristallstruktur der Verbindung LiNa[Ge4O9], Monatsh.Chem., 1969, vol. 100, no. 1, pp. 79–90.
Matveeva, R.G., Ilyukhin, V.V., and Belov, N.V., Crystal Structure of a Mixed Alkali Tetragermanate, Dokl. Akad.Nauk SSSR, 1973, vol. 213, no. 3, pp. 584–587.
Zemann, J., Isotypie zwischen Pharmakosiderit und zeolithishen Germanaten, Acta Crystallogr., 1959, vol. 12, no. 2, p. 252.
Iwata, Y., Koyano, N., Wada, M., and Sawada, W., On the Space Group of the Low Temperature Phase of LiNaGe4O9, Annu. Rep. Reactor Inst. Kyoto Univ., 1995, vol. 28, pp. 13–17.
Völlenkle, H., Wittmann, A., and Nowotny, H., Die Kristalstruktur des Lithiumenegermanats Li4Ge9O20, Monatsh. Chem., 1971, vol. 102, nos. 1–4, pp. 361–372.
Völlenkle, H., Wittmann, A., and Nowotny, H., Die Kristalstructure des Lithiumheptagermanats Li2Ge7O15, Monatsh. Chem., 1970, vol. 101, no. 1, pp. 46–56.
Greenberg, B.L. and Loiacono, G.M., Structure of Li4Ge5O12—a New Compound in the Li2O=GeO2 System, Acta Crystallogr., 1990, vol. 46, no. 10, pp. 2021–2026.
Cascales, C., Gutierrez-Puebla, E., Monge, M.A., and Ruiz-Valero, C., (NH4)2 Ge7O15: A Microporous Material Containing GeO4 and GeO6 Polyhedra in Nine-Rings, Angew. Chem., 1998, vol. 37, no. 1, pp. 129–131.
Behrens, E.A. and Clearfield, A., Titanium Silicates M3HTi4O4(SiO4)3 · 4H2O (M = Na+, K+) with Three-Dimensional Tunnel Structures for the Selective Removal of Strontium and Cesium from Wastewater Solutions, Microporous Mater., 1997, vol. 11, no. 1/2, pp. 65–75.
Dadachov, M.S. and Harrison, W.T.A., Synthesis and Crystal Structure of Na4 ((Ti O)4 (SiO4)3 · 6H2O, a Rhombohedrally Distorted Sodium Titanium Silicate Pharmacosiderite Analogue, J. Solid State Chem., 1997, vol. 134, no. 2, pp. 409–415.
Lobachev, A.N., Kuzmina, I.P., and Demianets, L.N., iet al., Uber die Rolle der Alkalimetalle beim Aufbau der Silicate und Germanate under Hydrothermalen Bedingungen, Krist. Tech., 1970, vol. 5, no. 2, pp. 425–432.
Tomilov, N.R., Samsonova, T.I., Poroshina, I.A., and Berger, A.S., Solid Phases in the Li2O–SiO2–H2O System, Zh. Neorg. Khim., 1978, vol. 23, no. 12, pp. 3333–3339.
Ilyushin, G.D. and Demianets, L.N., Hydrothermal Systems LiOHÐ(MeO2)–GeO2–H2O (Me—Ti, Sn, Zr, Hf): Phase Relations and Crystal-Chemical Correlations between Li,Me-and Li,Ge-Germanates, Joint 6th Int.Symp. on Hydrothermal Reactions & 4th Int. Conf. on Solvo-Thermal Reactions, Kochi, 2000, p. 2P-27.
Ilyushin, G.D. and Blatov, B.A., Crystal Chemistry of Zirconosilicates and Their Analogs: Topological Classi-fication of MT-Frameworks and Suprapolyhedral Invariants, Acta Crystallogr., 2002, vol. 58, no. 2, pp. 198–218.
Ilyushin, G.D., Dem'yanets, L.N., Ilyukhin, V.V., and Belov, N.V., Preparation of Analogs of Natural Minerals and Synthetic Phases in the Hydrothermal System NaOH–ZrO2–SiO2–H2O, Dokl. Akad. Nauk SSSR, 1983, vol. 271, no. 5, pp. 1133–1136.
Ilyushin, G.D. and Dem'yanets, L.N., Hydrothermal Synthesis of K2ZrSi6O15, K2ZrSi3O9, K2ZrSi2O7, and ZrSiO4 in the System KOH–ZrO2–SiO2–H2O, Neorg.Mater., 2002, vol. 38, no. 6, pp. 739–744 [Inorg. Mater. (Engl. Transl.), vol. 38, no. 6, pp. 612–616
Kennedy, G.C., Pressure–Volume–Temperature Relations in Water at Elevated Temperatures and Pressures, Am. J. Sci., 1950, vol. 248, p. 540.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Ilyushin, G.D., Dem'yanets, L.N. Phase Relations in the Systems LiOH–GeO2–H2O and LiOH–SiO2–H2O at 500°C and 0.1 GPa. Inorganic Materials 38, 700–708 (2002). https://doi.org/10.1023/A:1016244526014
Issue Date:
DOI: https://doi.org/10.1023/A:1016244526014