Abstract
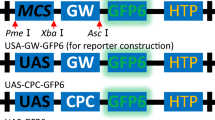
We have constructed a binary vector for Agrobacterium-mediated plant transformation, which has a multiple cloning site consisting of 13 hexanucleotide restriction sites, 6 octanucleotide restriction sites and 5 homing endonuclease sites. The homing endonuclease sites have the advantages to be extremely rare in natural sequences and to allow unidirectional cloning. We have also constructed a set of auxiliary vectors allowing the assembly of expression cassettes flanked by homing endonuclease sites. The expression cassettes assembled in these auxiliary vectors can be transferred into the binary vector with virtually no risk of cutting the vector within previously introduced sequences. This vector set is ideally suited for the construction of plant transformation vectors containing multiple expression cassettes and/or other elements such as matrix attachment regions. With this modular vector system, six different expression units were constructed in as many auxiliary vectors and assembled together in one plant transformation vector. The transgenic nature of Arabidopsis thaliana plants, transformed with this plant transformation vector, was assessed and the expression of each of the six genes was demonstrated.
Similar content being viewed by others
References
Barker, R.F., Idler, K.B., Thompson, D.V. and Kemp, J.D. 1983. Nucleotide sequence of the T-DNA region from Agrobacterium tumefaciens octopine Ti plasmid pTi15955. Plant Mol. Biol. 2: 335–350.
Bechtold, N., Ellis, J. and Pelletier, G. 1993. In planta Agrobacterium-mediated gene transfer by infiltration of adult Arabidopsis thaliana plants. C. R. Acad. Sci. 316: 1194–1199.
Becker, D., Kemper, E., Schell, J. and Masterson, R. 1992. New plant binary vectors with selectable markers located proximal to the left T-DNA border. Plant Mol. Biol. 20: 1195–1197.
Bevan, M. 1984. Binary Agrobacterium vectors for plant transformation. Nucl. Acids Res. 12: 8711–8721.
Binet, M.-N., Weil, J.-H. and Tessier, L.-H. 1991. Structure and expression of sunflower ubiquitin genes. Plant Mol. Biol. 17: 395–407.
Bradford, M.M. 1976. A rapid and sensitive method for the quantification of microgram quantities utilizing the principle of protein-dye binding. Anal. Biochem. 72: 248–254.
Clough, S.J. and Bent, A.F. 1998. Floral dip: a simplified method for Agrobacterium-mediated transformation of Arabidopsis thaliana. Plant J. 16: 735–743.
Colleaux, L., D'Auriol, L., Galibert, F. and Dujon, B. 1988. Recognition and cleavage site of the intron-encoded omega transposase. Proc. Natl. Acad. Sci. USA 85: 6022–6026.
Datla, R.S.S., Bekkaoui, F., Hammerlindl, J.K., Pilate, G., Dunstan, I.D. and Crosby, W.L. 1993. Improved high-level constitutive foreign gene expression using an AMV RNA4 untranslated leader sequence. Plant Sci. 94: 139–149.
de Majnik, J., Joseph, R.G., Tanner, G.J., Larkin, P.J., Djordjevic, M.A., Rolfe, B.G. and Weinman, J.J. 1997. A convenient set of vectors for expression of multiple gene combinations in plants. Plant Mol. Biol. Rep. 15: 134–140.
Depicker, A., Stachel, S., Dhaese, P., Zambryski, P. and Goodman, H.M. 1982. Nopaline synthase: transcript mapping and DNA sequence. J. Mol. Appl. Genet. 1: 561–573.
Eggermont, K., Goderis, I.J. and Broekaert, W.F. 1996. High-throughput RNA extraction from plant samples based on homogenisation by reciprocal shaking in the presence of a mixture of sand and glass beads. Plant Mol. Biol. Rep. 14: 273–279.
Gallie, D.R. and Walbot, V. 1992. Identification of the motifs within the tobacco mosaic virus 5?-leader responsible for enhancing translation. Nucl. Acids Res. 17: 4631–4638.
Gardner, R.C., Howarth, A.J., Hahn, P., Brown-Luedi, M., Shepperd, R.J. and Messing, J. 1981. The complete nucleotide sequence of an infectious clone of cauliflower mosaic virus by M13mp7 shotgun sequencing. Nucl. Acids Res. 9: 2871–2888.
Hajdukiewicz, P., Svab, Z. and Maliga, P. 1994. The small, versatile pPZP family of Agrobacterium binary vectors for plant transformation. Plant Mol. Biol. 25: 989–994.
Hellens, R., Mullineaux, P. and Klee, H. 2000. A guide to Agrobacterium binary Ti vectors. Trends Plant Sci. 5: 446–451.
Holmes-Davis, R. and Comai, L. 1998. Nuclear matrix attachment regions and plant gene expression. Trends Plant Sci. 3: 91–97.
Jefferson, R.A. 1987. Assaying chimeric genes in plants: the GUS gene fusion system. Plant Mol. Biol. Rep. 5: 387–405.
Jefferson, R.A. and Wilson, K.J. 1991. The GUS gene fusion system. In: S.B. Gelvin and R.A. Schilperoort (Eds.) Plant Molecular Biology Manual, Kluwer Academic Publishers, Dordrecht, Netherlands, pp. B14: 1–33.
Jefferson, R.A., Burgess, S. and Hirsh D. 1986. ?-glucuronidase from E. coli as a gene fusion marker. Proc. Natl. Acad. Sci. USA 83: 8447–8451.
Kooter, J.M., Matzke, M.A. and Meyer, P. 1999. Listening to silent genes: transgene silencing, gene regulation and pathogen control. Trends Plant Sci. 4: 340–347.
Liu, S., Hessel, A. and Sanderson, K.E. 1993. Genomic mapping with I-CeuI, an intron-encoded endonuclease specific for genes for ribosomal RNA, in Salmonella spp., Escherichia coli, and other bacteria. Proc. Natl. Acad. Sci. USA 90: 6874–6878.
Lonsdale, D.M., Moisan, L.J. and Harvey, A.J. 1995. pFC1 to pFC7: a novel family of combinatorial cloning vectors. Plant Mol. Biol. Rep. 13: 343–345.
Marshall, P. and Lemieux, C. 1992. The I-CeuI endonuclease recognizes a sequence of 19 base pairs and preferentially cleaves the coding strand of the Chlamydomonas moewusii chloroplast large subunit rRNA gene. Nucl. Acids Res. 20: 6401–6407.
Manners, J.M., Penninckx, I.A.M.A., Vermaere, K., Kazan, K., Brown, R.L., Morgan, A., Maclean, D.J., Curtis, M.D., Cammue, B.P.A. and Broekaert, W.F. 1998. The promoter of the plant defensin gene PDF1.2 from Arabidopsis is systemically activated by fungal pathogens and responds to methyl jasmonate but not to salicylic acid. Plant Mol. Biol. 38: 1071–1080.
McCormac, A.C., Elliott, M.C. and Chen, D.F. 1997. A flexible series of binary vectors for Agrobacterium-mediated plant transformation. Mol. Biotechnol. 8: 199–213.
Millar, A.J., Short, S.R., Hiratsuka, K., Chua, N.-H. and Kay, S.A. 1992. Firefly luciferase as a reporter of regulated gene expression in higher plants. Plant Mol. Biol. Rep. 10: 324–337.
Muscarella, D.E., Ellison, E.L., Ruoff, B.M. and Vogt, V.M. 1990. Characterization of I-Ppo, an intron-encoded endonuclease that mediates homing of a group I intron in the ribosomal DNA of Physarum polycephalum. Mol. Cell Biol. 10: 3386–3396.
Norrander, J., Kempe, T. and Messing, J. 1983. Construction of improved M13 vectors using oligodeoxynucleotide-directed mutagenesis. Gene 26: 101–106.
Osborn, R.W., De Samblanx, G.W., Thevissen, K., Goderis, I., Torrekens, S., Van Leuven, F., Attenborough, S., Rees, S.B. and Broekaert W.F. 1995. Isolation and characterization of plant defensins from seeds of Asteraceae, Fabaceae, Hippocastanaceae and Saxifragaceae. FEBS Lett. 368: 257–262.
Ow, D.W., Wood, K.V., DeLuca, M., de Wet, J.R., Helinski, D.R. and Howell, S.H. 1986. Transient and stable expression of the firefly luciferase gene in plant cells and transgenic plants. Science 234: 856–859.
Penninckx, I.A.M.A., Eggermont, K., Terras, F.R.G., Thomma, B.P.H.J., De Samblanx, G.W., Buchala, A., Métraux, J.P., Manners, J.M. and Broekaert, W.F. 1996. Pathogen-induced systemic activation of a plant defensin gene in Arabidopsis follows a salicylic-independent pathway. Plant Cell 8: 257–262.
Perler, F.B., Davis, E.O., Dean, G.E., Gimble, F.S., Jack, W.E., Neff, N., Noren, J.C., Thorner, J. and Belfort, M. 1994. Protein splicing elements: inteins and exteins: a definition of terms and recommended nomenclature. Nucl. Acids Res. 11: 1125–1127.
Sambrook, J., Fritsch, E.F. and Maniatis, T. 1989. Molecular Cloning: A Laboratory Manual, 2nd ed. Cold Spring Harbor Laboratory Press, Plainview, NY.
Sathasivan, K., Haughn, G.W. and Muria, N. 1990. Nucleotide sequence of a mutant acetolactate synthase gene from an imidazoline-resistant Arabidopsis thaliana var. Columbia. Nucl. Acids Res. 18: 2188.
Terras, F.R.G., Schoofs, H.M.E., De Bolle, M.F.C., Van Leuven, F., Rees, S.B., Vanderleyden, J., Cammue, B.P.A. and Broekaert, W.F. 1992. Analysis of two novel classes of plant antifungal proteins from radish (Raphanus sativus L.) seeds. J. Biol. Chem. 267: 15301–15309.
Terras, F.R.G., Eggermont, K., Kovaleva, V., Raikhel, N.V., Osborn, R.W., Kester, A., Rees, S.B., Torrekens, S., Van Leuven, F., Vanderleyden, J., Cammue, B.P.A. and Broekaert, W.F. 1995. Small cysteine-rich antifungal proteins from radish: their role in host defense. Plant Cell 7: 573–588.
Ñberlacker, B. and Werr, W. 1996. Vectors with rare-cutter restriction enzyme sites for expression of open reading frames in transgenic plants. Mol. Breed. 2: 293–295.
Verdaguer, B., de Kochko, A., Fux, C.I., Beachy, R.N. and Fauquet, C. 1998. Functional organization of the cassava vein mosaic virus (CsVMV) promoter. Plant Mol. Biol. 37: 1055–1067.
White, J., Chang, S.P. and Bibb, M.J. 1989. A cassette containing the bar gene of Streptomyces hygroscopicus: a selectable marker for plant transformation. Nucl. Acids Res. 18: 1062.
Zambryski, P., Joos, H., Genetello, C., Leemans, J., Van Montagu, M. and Schell, J. 1983. Ti plasmid vectors for the introduction of DNA into plant cells without alteration of their normal regeneration capacity. EMBO J. 2: 2143–2150.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Goderis, I.J., De Bolle, M.F., François, I.E. et al. A set of modular plant transformation vectors allowing flexible insertion of up to six expression units. Plant Mol Biol 50, 17–27 (2002). https://doi.org/10.1023/A:1016052416053
Issue Date:
DOI: https://doi.org/10.1023/A:1016052416053