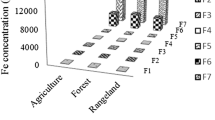
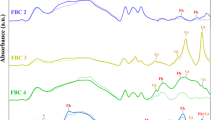
Abstract
The most effective and common Fe fertilisers in general are EDDHA and EDDHMA Fe chelates because they are highly stable ferric complexes in neutral and alkaline solutions. EDDHSA and EDDCHA iron chelates were introduced in the market recently. Commercial Fe chelates have two Fe fractions, chelated Fe and non-chelated Fe. The latter is bonded to by-products produced during the synthesis of the chelating agent. The effectiveness of Fe chelates depends on their ability to maintain Fe in the soil solution despite simultaneous equilibrium of Fe chelate with many cations, such as Ca2+. The main aim of this work was to test the possible agricultural use of EDDHSA and EDDCHA Fe chelates. The pH-Ca2+ effect on soluble and chelated Fe (pH ranging from 2 to 12) and the interaction of Fe chelates with soils and soil phases (ferrihydrite, acid peat, calcium carbonate and Ca montmorillonite) are presented. The results demonstrated that EDDHA, EDDHMA, EDDHSA and EDDCHA in solution remain fully associated with Fe from pH 4 to 9 despite competition with Ca. Among soil materials, ferrihydrite and acid peat retain both chelated and non-chelated Fe to the greatest extent. The type of chelating agent is a factor that affects chelated Fe availability in soil. FeEDDHA and FeEDDHMA were retained more by soil surfaces than FeEDDHSA and FeEDDCHA. Commercial Fe chelates present a large amount of soluble, non-chelated Fe and make Cu soluble in soils, which may be due to non-chelated Fe being displaced by Cu.
Similar content being viewed by others
References
Ahrland S, Dahlgren A and Persson I 1990 Stabilities and hydrolysis of some Fe (III) and manganese (III) complexes with chelating ligands. Acta Agric. Scand. 40, 101-111.
Álvarez-Fernández A, Gárate A and Lucena J J 1997 Interaction of Fe chelates with several soil materials and with a soil standard. J. Plant Nutr. 20, 559-572.
Álvarez-Fernández A, Yunta-Mezquita F, Sierra M A and Lucena J J. 2002. Quality of European Fe chelates. The case of Spain. In 10th International Symposium on Iron nutrition and Interactions in Plants. Eds. Z Rengel and M Grusak. (Forthcoming). Kluwer Academic Publishers, Dordrecht, The Netherlands.
Assaad F F and Awad F 1981 The stability of Fe chelates in calcareous soils. Z. Pflanzenernaehr. Bodenkd. 144, 77-86.
Bannochie J and Martell A E 1989 Affinities of racemic and meso forms of N,N'-ethylenebis[2-(o-hydroxyphenyl)glycine] for divalent and trivalent metal ions. J. Am. Chem. Soc. 111, 4735-4742.
Bolt G H and Warkentin B P 1958 The negative adsorption of anions by clay suspensions. Kolloid Z. 156, 41-46.
Boxma R 1981 Effect of pH on the behaviour of various Fe chelates in sphagnum (moss) peat. Commun. Soil Sci. Plant Anal. 12, 755-763.
Brunauer S, Emmeth P H and Teller E 1938 Adsorption of gases in multimolecular layers. J. Am. Chem. Soc. 60, 309-319.
Hernández-Apaolaza L 1997 Determinación de quelatos férricos de uso agrícola. Aplicación al estudio de su adsorpción por materiales edáficos. PhD Thesis. Universidad Autónoma de Madrid, Spain.
Hill-Cottingham D G and Lloyd-Jones C P 1957 Behaviour of Fe chelates in calcareous soils. I. Laboratory experiments with Fe-EDTA and Fe-HEEDTA. Plant Soil 8, 263-274.
Hill-Cottingham D G and Lloyd-Jones C P 1958 Behaviour of Fe chelates in calcareous soils. II. Laboratory experiments with some further chelating agents. Plant Soil 9, 189-201.
Lindsay W L 1979 Chemical Equilibria in Soils. Ed. John Wiley and sons. N.Y., USA.
Lindsay W L, Norvell W A 1978 Development of a DTPA soil test for zinc, iron, manganese and copper. Soil Sci. Soc. Am. J. 42: 421-428.
Liñán C 1999 Correctores de carencias de hierro. In Vademecum de productos fitosanitarios y nutricionales. pp 360-369. Agrotécnicas, S.L., Madrid, Spain.
Lucena J J, Barak P and Hernández-Apaolaza L 1996 Isocratic ionpair high-performance liquid chromatographic method for the determination of various Fe(III) chelates. J. Chromatogr. A 727, 253-264.
Lucena J J, Gárate A and Carpena O 1988 Theoretical and practical studies on chelate-Ca-pH system in solution. J. Plant Nutr. 11, 1051-1061.
Lucena J L, Manzanares M and Gárate A 1992 Comparative study of the efficacy of commercial Fe chelates using a new test. J. Plant Nutr. 15, 1995-2006.
MAPA Ministry of Agriculture, Fisheries and Food 1994 Official Methods of Analysis (In Spanish). Volume III. 205-324.
Norvell W A 1991 Reactions of metal chelates in soils and nutrient solutions. In Micronutrients in Agriculture, 2nd edn. SSSA Book Series, no 4. Eds. Mortvedt J J, Cox F R, Shuman L M and Welch R M. pp. 187-227. Soil Science Society of America, Madison, Wisconsin, USA.
Norvell W A and Lindsay W L 1969 Reactions of EDTA complexes of iron, Zn, Mn, and Cu with soils. Soil Sci. Am. Proc. 33, 86-91.
Norvell W A and Lindsay W L 1972 Reactions of DTPA chelates of iron, zinc, copper and manganese with soils. Soil Sci. Soc. Am. Proc. 36, 778-783.
Sierra M A, Gómez-Gallego M, Alcázar-Romero R, Lucena J J, Álvarez-Fernández A and Yunta-Mezquita F 2000 Nuevo procedimiento para la preparación de ácidos bis(2-hidroxiaril) aminoacéticos utilizando agentes de transferencia de cianuro. Spaninsh Patent number P2000016002.
Soltanpour P N and Schwab A P 1977 A new soil test for simultaneous extraction of macro-and micro-nutrients in alkaline soils. Commun. Soil Sci. Plant Anal. 8, 195-207.
Wallace A, Mueller R T, Lunt O R, Ashcroft R T and Shannon L M 1955 Comparisons of five chelating agents in soils, in nutrient solutions, and in plant responses. Soil Sci. 80, 101-108.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Álvarez-Fernández, A., Sierra, M.A. & Lucena, J.J. Reactivity of synthetic Fe chelates with soils and soil components. Plant and Soil 241, 129–137 (2002). https://doi.org/10.1023/A:1016012513044
Issue Date:
DOI: https://doi.org/10.1023/A:1016012513044