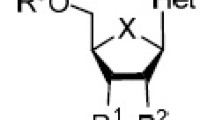Abstract
The structural requirements for the antiviral activity of pyrimidine nucleosides against HIV-1 virus was evaluated with the Hansch SAR analysis. Antiviral activity is best related to the hydrophobicity and steric (L and B3) properties of the substituent at the C5 of pyrimidine ring. Further, the antiviral activity is related to B4 of the substituent at position 3' of the sugar ring with a positive slope. The activity of both uracil and cytosine derivatives can be related to their structure by the same equations, which indicates that the SARs are similar in these two groups of congeners. These results suggest that compounds with a small substituent at the 5 position of the pyrimidine ring and a flat substituent at the 3' position of the sugar ring will be the most active compounds against HIV-1 virus.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
REFERENCES
H. Mitsuya, K. J. Weinhold, P. A. Furman, M. H. St. Clair, S. N. Lehrman, R. L. Gallo, D. Bolognesi, D. W. Barry, and S. Broder. 3′-Azido, 3′-deoxythymidine (BW A509U): An antiviral agent that inhibits the infectivity and cytopathic effects of human T cell lymphotropic virus III/lymphadenophath-associated virus in-vitro. Proc. Natl. Acad. Sci. USA 82:7096–7100 (1985).
P. A. Furman and D. W. Barry. Spectrum of antiviral activity and mechanism of action of zidovudine. Am. J. Med. 85:176–181 (1988).
D. D. Richman. Results of continued monitoring of participants in the placebo-controlled trial of zidovudine for serious human immunodeficiency virus infection. Am. J. Med. 85:208–213 (1988).
K. C. Chu, R. F. Schinazi, M. K. Ahn, G. V. Ullas, and Z. P. Gu. Structure-activity relationships of pyrimidine nucleosides as antiviral agents for human immunodeficiency virus type I in peripheral blood mononuclear cells. J. Med. Chem. 32:612–617 (1989).
T.-S. Lin, J.-Y. Guo, R. F. Schinazi, C. K. Chu, J.-N. Xiang, and W. H. Prusoff. Synthesis and antiviral activity of various 3′-azido analogues of pyrimidine deoxyribonucleosides against human immunodeficiency virus (HIV-1, HTLV-III/LAV). J. Med. Chem. 31:336–340 (1988).
C. Hansch. In C. J. Cavallito (ed.). Structure Activity Relationships, Pergamon Press, Oxford, 1973, pp. 75–165.
A. Verloop, W. Hoogenstraaten, and J. Tipker. Development and application of new steric substituent parameters in drug design. In E. J. Ariens (ed.), Drug Design, Vol. 7, Academic Press, New York, 1976, pp. 165–207.
S. H. Unger. Consequences of the Hansch paradigma for the pharmaceutical industry. In E. J. Ariens (eds.), Drug Design, Vol. 9, Academic Press, New York, 1980, pp. 48–119.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Mahmoudian, M. Quantitative Structure–Activity Relationships (QSARs) of Pyrimidine Nucleosides as HIV-1 Antiviral Agents. Pharm Res 8, 43–46 (1991). https://doi.org/10.1023/A:1015822105022
Issue Date:
DOI: https://doi.org/10.1023/A:1015822105022