Abstract
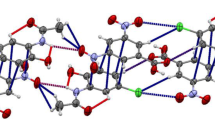
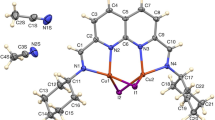
p-Bromo-N-(p-cyanobenzylidene)aniline is triclinic, has space group P \(\overline 1\), at 173 K, with a = 7.3892(15), b = 12.087(2), c = 7.0283(14) Å, α = 105.24(2), β = 98.11(2), γ = 98.89(2)°, Z = 2, V = 587.4(2) Å3. p-Chloro-N-(p-cyanobenzylidene)aniline is triclinic, space group P \(\overline 1\), at 173 K, with a = 7.317(2), b = 12.165(3), c = 6.990(2) Å, α = 104.02(2), β = 100.96(2), γ = 100.76(2)°, Z = 2, V = 574.7(3) Å3. p-Cyano-N-(p-bromobenzylidene)aniline is orthorhombic, has space group P212121, at 173 K, with a = 10.333(3), b = 4.732(3), c = 23.526(5) Å, Z = 4, V = 1150.3(8) Å3. p-Cyano-N-(p-chlorobenzylidene)aniline is orthorhombic, has space group P212121, at 173 K, with a = 10.161(2), b = 4.713(2), c = 23.509(5) Å, Z = 4, V = 1125.8(4) Å3. In the two cyanobenzylidene compounds there are chains of molecules held together by CN······X interactions. The chains form two-dimensional sheets similar to those in the iodo analog, but with a different stacking arrangement between the sheets. In the two halobenzylidene compounds there are no CN······X contacts; rather there are short X······X and C–H······NC contacts.
Similar content being viewed by others
References
Ojala, C.R.; Ojala, W.H.; Gleason, W.B.; Britton, D. J.Chem. Crystallogr. 1999, 29, 27.
Bernstein, J.; Izak, I. J.Chem.Soc., Perkin II 1976, 429.
Bernstein, J.; Schmidt, G.M.J. J.Chem.Soc., Perkin II 1972, 951.
Bar, I.; Bernstein, J. Acta Crystallogr., Sect.B 1982, 38, 121.
Bar, I.; Bernstein, J. Acta Crystallogr., Sect.B 1977, 33, 1738.
Bernstein, J.; Bar, I.; Christensen, A. Acta Crystallogr., Sect.B 1976, 32, 1609.
Bar, I.; Bernstein, J. Acta Crystallogr., Sect.B 1983, 39, 266.
Haller, K.J.; Rae, A.D.; Heerdegen, A.P.; Hockless, D.C.R.; Welberry, T.R. Acta Crystallogr., Sect.B 1995, 51, 187.
Flack, H.D. Acta Crystallogr., Sect.A 1983, 39, 876.
North, A.C.T.; Phillips, D.C.; Mathews, F.S. Acta Crystallogr., Sect.A 1968, 24, 351.
Gilmore, C.J. J.Appl.Crystallogr. 1984, 17, 42.
Beurskens, P.T. DIRDIF: Direct Methods for Difference Structures; Technical Report 1984/1; Crystallography Laboratory: Toernooiveld, Nijmegen, The Netherlands, 1984.
Molecular Structure Corp. TEXSAN, TEXRAY Structure Analysis Package; MSC: The Woodlands, TX, 1985.
Sheldrick, G.M. SADABS, Absorption Correction Program; University of G¨ottingen: Germany, 1996.
Blessing, R.H.Acta Crystallogr., Sect.A 1995, 51, 33
Sheldrick, G.M. SHELXTL, Version 5.0; Siemens Analytical X-ray Instruments, Inc.: Madison, WI, 1994.
Duisenberg, A.J.M. J.Appl.Crystallogr. 1992, 25, 92.
Pauling, L. The Nature of the Chemical Bond, 3rd ed.; Cornell University Press: Ithaca, 1960; p 260.
Khanna, R.; Welberry, T.R. Acta Crystallogr., Sect.A 1990, 46, 975, and references therein.
Bent, H.A. Chem.Rev. 1968, 68, 587.
Sakurai, T.; Sundaralingam, M.; Jeffrey, G.A. Acta Crystallogr. 1963, 16, 354.
Ramasubbu, N.; Parthasarathy, R.; Murray-Rust, P. J.Am.Chem. Soc. 1986, 108, 4308.
Desiraju, G.R. Crystal Engineering; Elsevier: Amsterdam, 1989; pp 179–185.
Nyburg, S.C.; Faerman, C.H. Acta Crystallogr., Sect.B 1986, 41, 274.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Ojala, C.R., Ojala, W.H., Gleason, W.B. et al. The crystal structures of p-halo-N-(p-cyanobenzylidene)aniline and p-cyano-N-(p-halobenzylidene)aniline (halo = bromo and chloro). Journal of Chemical Crystallography 31, 377–386 (2001). https://doi.org/10.1023/A:1015799223882
Issue Date:
DOI: https://doi.org/10.1023/A:1015799223882