Abstract
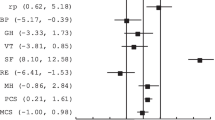
Objective: To assess the equivalence of English and Chinese versions of the SF-36. Methods: Using a crossover design with block randomisation and stratification by age, identical English or Chinese questionnaires containing the English (UK) and Chinese (HK) SF-36 versions were administered 3–16 days apart to 168 free-living, bilingual, ethnic Chinese volunteers in Singapore. Item level equivalence of both versions was assessed by comparing item means and orderings within each scale. Scale level equivalence was assessed by comparing internal consistency (Cronbach's α), results of factor analysis and mean scale scores (using paired t-tests and intra-class correlations). Results: Item and scale level comparisons supported the equivalence of both versions. For both the versions, item means, item ordering and Cronbach's α were similar, and factor analysis yielded two factors with similar factor loadings. There was no clinically important difference in mean scale scores for seven of eight scales, and intra-class correlations were excellent/good for five scales (0.69–0.77) and moderate for three scales (0.55–0.57). Conclusion: English (UK) and Chinese (HK) SF-36 versions are equivalent in bilingual Singapore Chinese. Our data suggest that SF-36 scores from English- and Chinese-speaking subjects may be combined in studies using the SF-36, increasing the power and representativeness of such studies.
Similar content being viewed by others
References
Ware JE. SF-36 Health Survey: Manual and interpretation guide. Boston: The Health Institute, New England Medical Center 1993; 3: 20-21.
Ware JE Jr, Gandek B. Overview of the SF-36 Health Survey and the International Quality of Life Assessment (IQOLA) Project. J Clin Epidemiol 1998; 51: 903-912.
Thumboo J, Fong KY, Machin D, et al. A community based study of scaling assumptions and construct validity of the English (UK) and Chinese (HK) SF-36 in Singapore. Qual Life Res 2001; 10: 175-188.
Guillemin F, Bombardier C, Beaton D. Cross-cultural adaptation of health-related quality of life measures: Literature review and proposed guidelines. J Clin Epidemiol 1993; 46: 1417-1432.
Ware JE, Gandek B. Methods for testing data quality, scaling assumptions, and reliability: The IQOLA project approach. J Clin Epidemiol 1998; 51: 945-952.
Ware JE, Gandek B. Methods for validating and norming translations of health status questionnaires: The IQOLA project approach. J Clin Epidemiol 1998; 51: 953-959.
Lam CL, Gandek B, Ren XS, Chan MS. Tests of scaling assumptions and construct validity of the Chinese (HK) version of the SF-36 Health Survey. J Clin Epidemiol 1998; 51: 1139-1147.
Senn S. Cross-over Trials in Clinical Research. 1st ed. New York: John Wiley & Sons, 1993.
Ruta DA, Abdalla MI, Garratt AM, Coutts A, Russell IT. SF-36 health survey questionnaire: I. Reliability in two patient based studies. Qual Health Care 1994; 3: 180-185.
Ware JE, Sherbourne CD. The MOS 36-Item Short Form Health Survey (SF-36) I: Conceptual framework and item selection. Med Care 1991; 30: 473-483.
McHorney C, Ware JEJ, Raczek A. The MOS 36-Item Short-Form Health Survey (SF-36): II. Psychometric and clinical tests of validity in measuring physical and mental health constructs. Med Care 1993; 31: 247-263.
McHorney C, Ware JE J, Lu J, Sherbourne C. The MOS 36-item Short-Form Health Survey (SF-36): III. Tests of data quality, scaling assumptions, and reliability across diverse patient groups. Med Care 1994; 32: 40-66.
Brazier JE, Harper R, Jones NMB, O'Cathain A, Thomas KJ, Usherwood T et al. Validating the SF-36 health survey questionnaire: New outcome measure for primary care. Br Med J 1992; 305: 160-164.
Dupont WD, Plummer WD. Power and sample size calculations. A review and computer programme. Controlled Clin Trials 1990; 11: 116-128.
Thumboo J, Fong KY, Ng TP, et al. The SF-36 is sensitive to changes in quality of life in SLE patients (Abstract). Arthritis Rheum 1998; 41(9S): S225.
Bland JM, Altman DG. Statistics Notes: Cronbach's alpha. Br Med J 1997; 314: 572.
Sharma S. Applied Multivariate techniques. New York: John Wiley and Sons, 1996; 90-143.
Bland JM, Altman DG. Statistical methods for assessing agreement between two methods of clinical assessment. Lancet 1986; 1: 307-310.
Fleiss JL. The Design and Analysis of Clinical Experiments. New York: John Wiley and Sons, 1986.
Chan DW. The Chinese version of the general health questionnaire: Does language make a difference? Psychol Med 1985; 15: 147-155.
Krongrad A, Perczek RE, Burke MA, Granville LJ, Lai H, Lai SH. Reliability of Spanish translations of select urological quality of life instruments. J Urol 1997; 158: 493-496.
Ware JE, Kosinski M. SF-36 Health Survey (version 2.0) Technical Note. Boston. MA: Health assessment lab, September 20, 1996 (updated September 27, 1997).
Hunt SM, Alonso J, Bucquet D, Niero M, Wiklund I, McKenna S. Cross-cultural adaptation of health measures. Health Policy 1991; 19: 33-44.
Thumboo J, Fong KY, Machin D, et al. Does Being Bilingual in English and Chinese Influence Responses to Quality of Life Scales? Med Care 2002; 40: 105-112.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Thumboo, J., Fong, KY., Chan, SP. et al. The equivalence of English and Chinese SF-36 versions in bilingual Singapore Chinese. Qual Life Res 11, 495–503 (2002). https://doi.org/10.1023/A:1015680029998
Issue Date:
DOI: https://doi.org/10.1023/A:1015680029998