Abstract
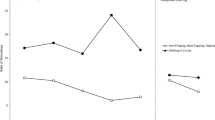
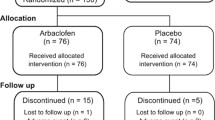
Secretin has been proposed as a treatment alternative for autistic spectrum disorders, but empirical support is lacking. A double-blind placebo-controlled study examined the effect of a single dose of synthetic human secretin on aberrant behavior. Parent and teacher data from the Aberrant Behavior Checklist for eight male children were analyzed for reliable change in a clinical replication series. By parent and teacher report, the majority of change occurred either on the placebo trial or reflected deterioration subsequent to secretin infusion. Repeated-measures multivariate analysis of variance results were similar. Results are consistent with other studies, suggesting that secretin may not be an effective treatment option.
Similar content being viewed by others
REFERENCES
Aman, M. G., & Singh, N. N. (1986). Aberrant Behavior Checklist manual. East Aurora, NY: Slosson.
Barlow, D. H., & Hersen, M. (1984). Single case experimental designs: Strategies for studying behavior change (2nd ed.). Oxford: Pergamon.
Chez, M. G., Buchanan, C. P., Bagan, B. T., Hammer, M. S., McCarthy, K. S., Ovrutskaya, I., Nowinski, C. V., & Cohen, Z. S. (2000). Secretin and autism: A two-part clinical investigation. Journal of Autism & Developmental Disorders, 30, 87–94.
D'Eufemia, P., Celli, M. Finocchiario, L., Pacifico, L., Viozzi, L., Zaccagnini, M., Cardi E., & Giardini, O (1996). Abnormal intestinal permeability in children with autism. Acta Paediatrica, 85, 1076–1079.
Horvath, K., Stefanatos, G., Sokolski, K. N., Watchel, R., Nabors, L., & Tildon, J. T. (1998). Improved social and language skills after secretin administration in patients with autistic spectrum disorders. Journal of the Association for Academic Minority Physicians, 9, 9–15.
Horvath, K., Zielke, R. H., Collins, R. M., Rabsztyn A., Medeiros, L. A., & Perman, J. (August 2000). Secretin improves the intestinal permeability in autistic children. Paper presented at the World Congress of Pediatric Gastroenerology, Boston, MA.
Jacobson, N. S., & Truaxx, P. (1991). A statistical approach to defining meaningful change in psychotherapy research. Journal of Consulting and Clinical Psychology, 59, 12–19.
Owley, T., Steele, E., Cosello, C., Risi, S., McKaig, K., Lord, C., Leventhal, B. L., & Cook, E. H. (1999). A double-blind, placebo-controlled trial of secretin for the treatment of autistic disorder. Medscape General Medicine, 1, 1–9.
Richman, D. M., Reese, R. M., & Daniels, D. (1999). Use of evidence-based practice as a method for evaluating the effects of secretin on a child with autism. Focus on Autism & Other Developmental Disabilities, 14, 204–211.
Sandler, A. D. & Bodfish, J. W. (2000). Placebo effects in autism: Lessons from Secretin. Developmental and Behavioral Pediatrics, 21, 347–348.
Sandler, A. D., Sutton, K. S., DeWeese, B. S., Girardi, M. A., Sheppard, V., & Bodfich, J. W. (1999). Lack of benefit of a single dose of synthetic human secretin in the treatment of autism and pervasive developmental disorder. The New England Journal of Medicine, 431, 1801–1806.
Schopler, E., Reichler, R., & Renner, B. R. (1988). Manual for the Childhood Autism Rating Scale. Los Angeles, CA: Western.
Schutt, C. E. (1998). Secretin & autism. Naarrative [online], 4.
Weiss, M. J. (1999). Differential rates of skill acquisition and outcomes of early intensive behavioral intervention for autism. Behavioral Interventions, 14, 3–22.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Carey, T., Ratliff-Schaub, K., Funk, J. et al. Double-Blind Placebo-Controlled Trial of Secretin: Effects on Aberrant Behavior in Children with Autism. J Autism Dev Disord 32, 161–167 (2002). https://doi.org/10.1023/A:1015493412224
Issue Date:
DOI: https://doi.org/10.1023/A:1015493412224