Abstract
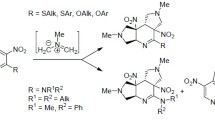
Unusual cascade cycloaddition reactions with dimethylhydrazones of α-trimethylsilyloxyalkylacroleins acting as azadienes were found. Depending on the nature of the dienophile, the reactions give either fused heterocyclic compounds (when two consecutive Diels—Alder reactions take place) or azabicyclic compounds, resulting from the [2+4]- and [2+3]-cycloaddition cascade.
Similar content being viewed by others
References
D. L. Boger, Chem. Rev., 1986, 86, 781; D. L. Boger and S. W. Weinreb, Hetero Diels-Alder Methodology in Organic Synthesis, Academic Press, New York, 1987.
E. Pascual-Alfonso, C. Avendano, and J. C. Menendez, Synlett, 2000, 205.
J. Barluenga and M. Tomas, Adv. Heterocycl. Chem., 1993, 189, 1.
H. Waldman, Synthesis, 1994, 535.
N. P. Tsvetkov, V. E. Vakhmistrov, A. B. Koldobsky, and V. N. Kalinin, Izv. Akad. Nauk, Ser. Khim., 1999, 1707 [Russ. Chem. Bull., 1999, 48, 1685 (Engl. Transl.)].
A. B. Koldobsky, V. E. Vakhmistrov, and V. N. Kalinin, Dokl. Akad. Nauk, 1996, 346, 771 [Dokl. Chem., 1996 (Engl. Transl.)].
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Tsvetkov, N.P., Vakhmistrov, V.E., Koldobsky, A.B. et al. Cascade cycloaddition reactions involving dimethylhydrazones of α-substituted acroleins. Russian Chemical Bulletin 51, 326–331 (2002). https://doi.org/10.1023/A:1015468013195
Issue Date:
DOI: https://doi.org/10.1023/A:1015468013195