Abstract
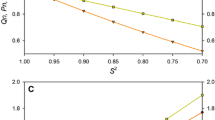
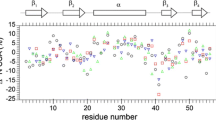
The heteronuclear 15N-NOE experiment was extensively tested with respect to statistical and systematic experimental error. The dependence of signal intensity in the NOE experiment and in the reference experiment on the saturation and relaxation time was experimentally investigated. The statistics of the experimental values were accessed by numerous repetitions of identical set-ups. As a model system a protein of typical size for NMR studies was chosen, i.e., a 120 amino acid residues containing fragment of the F-actin binding gelation factor (ABP-120). The fragment exhibits fast dynamics that are accessible with the 15N-NOE experiment with various amplitudes. The results of this study show that commonly used parameters are only adequate for accurate measurement of motions with moderate amplitude. Highly flexible parts require longer delay times and thus more experimental time than commonly used. On the other hand, a qualitative or semi-quantitative assessment of a protein's mobility on fast times scales can be obtained from rapidly recorded experiments with unusual short delay times. The findings of this study are of equal importance for highly accurate measurement of the 15N-NOE as well as for quick identification of mobile or even unstructured residues/parts of a protein.
Similar content being viewed by others
References
Bronstein, I.N. and Semendjajew, K.A. (1957) Taschenbuch der Mathematik, Teubner, Leipzig.
Buck, M., Schwalbe, H. and Dobson, Ch.M. (1996) J. Mol. Biol., 257, 669–683.
Cieslar, C., Ross, A., Zink, T. and Holak, T.A. (1993) J. Magn. Reson., 101, 97–101.
Clore, G.M., Driscoll, P.C., Wingfield, P.T. and Gronenborn, A.M. (1990) Biochemistry, 27, 7387–7401.
Dayie, K.T., Wagner, G. and Lefèvre, J.-F. (1996) In Dynamics and the Problem of Recognition in Biological Macromolecules Jardetzky, O. and Lefèvre, J.-F. (Eds.), NATO ASI Series A, Vol. 288, Plenum Press, New York, NY.
Farrow, N.A., Muhandiram, R., Singer, A.U., Pascal, S.M., Kay, C.M., Gish, G., Shoelson, S.E., Pawson, T., Foreman-Kay, J.D. and Kay, L.E. (1994) Biochemistry, 33, 5984–6003.
Fischer, M.W.F., Majumdar, A. and Zuiderweg, E.R.P. (1998) Prog. NMR Spectrosc., 33, 207–272.
Fucini, P., Renner, C., Herberholt, C., Noegel, A.A. and Holak, T.A. (1997) Nat. Struct. Biol., 4, 223–230.
Fushman, D., Cahill, S. and Cowburn, D. (1997) J. Mol. Biol., 266, 173–194.
Fushman, D., Tjandra, N. and Cowburn, D. (1999) J. Am. Chem. Soc., 121, 8577–8582.
Grzesiek, S. and Bax, A. (1993) J. Am. Chem. Soc., 115, 12593–12594.
Kay, L.E. (1998) Biochem. Cell Biol., 76, 145–152.
Kay, L.E., Torchia, D.A. and Bax, A. (1989) Biochemistry, 28, 8972–8979.
Lefèvre, J.-F., Dayie, K.T., Peng, J.W. and Wagner, G. (1996) Biochemistry, 35, 2674–2686.
Li, Y.-C., Montelione, G.T. (1994) J. Magn. Reson., B105, 45–51.
Markley, J.L., Horsley, W.J. and Klein, M.P. (1971) J. Chem. Phys., 55, 3604–3605.
Mühlhahn, P., Zweckstetter, M., Georgescu, J., Ciosto, C., Renner, C., Lanzendörfer, M., Lang, K., Ambrosius, D., Baier, M., Kurth, R. and Holak, T.A. (1998) Nat. Struct. Biol., 5, 682–686. 33
Orekhov, V.Y., Pervushin, K.V., Korzhnev, D.M. and Arsieniev, A.S. (1995) J. Biomol. NMR, 6, 113–122.
Palmer, A.G. (1997) Curr. Opin. Strut. Biol., 7, 732–737.
Papavoine, C.H.M., Remerowski, M.L., Horstink, L.M., Kronings, R.N.H., Hilbers, C.W. and van der Ven, F.J.M. (1997) Biochemistry, 36, 4015–4026.
Peng, J.W. and Wagner, G. (1992) Biochemistry, 31, 8571–8586.
Renner, C.and Holak, T.A. (2000) J. Magn. Reson., 145, 192–200.
Renner, C. and Holak, T.A. (2001) Eur. J. Biochem., 268, 1058–1065.
Renner, C., Baumgartner, R., Noegel, A.A. and Holak, T.A. (1998) J. Mol. Biol., 283, 221–229.
Skelton, N.J., Palmer III, A.G., Akke, M., Kördel, J., Rance, M. and Chazin, W.J. (1993) J. Magn. Reson., B102, 308–332.
Smith, G.M., Yu, L.P. and Domingues, D.J. (1987) Biochemistry, 26, 2202–2207.
Tessari, M., Mulder, F.A.A., Boelens, R. and Vuister, G.W. (1997) J. Magn. Reson., 127, 128–133.
Tjandra, N., Szabo, A. and Bax, A. (1996) J. Am. Chem. Soc., 118, 6986–6991.
Viles, J.H., Donne, D., Kroon, G., Prusinger, S.B., Cohen, F.E., Dyson, H.J. and Wright, P.E. (2001) Biochemistry, 40, 2743–2753.
Zheng, Z., Czaplicki, J. and Jardetzky, O. (1995) Biochemistry, 34, 5212–5223.
Zink, T., Ross, A., Lüers, K., Cieslar, C., Rudolph, R. and Holak, T.A. (1994) Biochemistry, 33, 8453–8463.
Author information
Authors and Affiliations
Corresponding author
Electronic Supplementary Material
Rights and permissions
About this article
Cite this article
Renner, C., Schleicher, M., Moroder, L. et al. Practical aspects of the 2D 15N-{1H}-NOE experiment. J Biomol NMR 23, 23–33 (2002). https://doi.org/10.1023/A:1015385910220
Issue Date:
DOI: https://doi.org/10.1023/A:1015385910220