Abstract
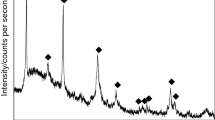
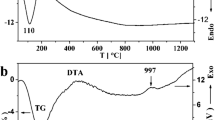
Forsterite MgSiO4 and enstatite MgSiO3 were synthesized by two different aqueous processes. TEOS was directly hydrolyzed in aqueous solutions of magnesium nitrate, giving solutions of magnesium nitrate and silicic acid. For the first process these solutions were spray-dried and the powders heat treated to decompose the nitrate; and for the second one they were precipitated in a solution of ethylenediamine as a base, the resulting precipitate was filtered, washed and dried. Spray-dried or precipitated, no specific thermal event was detected by thermal analysis for the crystallization of forsterite (500–1000°C) while a strong and sharp exothermic peak traduced the crystallization of enstatite at 800°C. Very minor secondary phases could be detected by X-ray diffraction up to 1200°C for the spray-dried powders, while the precipitated powders presented a higher chemical homogeneity, but much care had to be taken for a quantitative precipitation. As some minor secondary phases like SiO2 or some polymorphs of MgSiO3 could be not detected by XRD up to 1300°C, higher thermal treatments were necessary to control the purity or the desired phase.
Similar content being viewed by others
References
C.J. Brinker and G.W. Scherer, Sol-Gel Science: The Physics and Chemistry of Sol-Gel Processing (Academic Press, San Diego, 1990).
Y. Narendar and G.L. Messing, Catal. Today 35, 247 (1997).
M.B.D. Mitchell, D. Jackson, and P.F. James, J. Sol-Gel Sci. & Technol. 13, 359 (1998).
T. Ban, Y. Ohya, and Y. Takahashi, J. Am. Ceram. Soc. 82, 22 (1999).
A. Kazakos, S. Komarneni, and R. Roy, Mat. Lett. 9, 405 (1990).
N.P. Bansal, J. Am. Ceram. Soc. 71, 666 (1988).
J.M. Burlitch, M.L. Beeman, B. Riley, and D.L. Kohlstedt, Chem. Mat. 3, 692 (1991).
S.A. Jones, J.M. Burlitch, J.C. Duchamp, and T.M. Duncan, J. Sol-Gel Sci. & Technol. 15, 201 (1999).
M.H.E. Martin, C.K. Ober, C. Hubbard, W.D. Porter, and O.B. Cavin, J. Am. Ceram. Soc. 75, 1831 (1992).
M.B.D. Mitchell, D. Jackson, and P.F. James, J. Sol-Gel Sci. & Technol. 15, 211 (1999).
L.N. Echeverría, J. Non-Cryst. Solids 147/148, 559 (1992).
G.L. Messing, S.-C. Zhang, and G. Jayanthi, J. Am. Ceram. Soc. 76, 2707 (1993).
A. Douy and M. Gervais, J. Am. Ceram. Soc. 83, 70 (2000).
V. Montouillout, D. Massiot, A. Douy, and J.P. Coutures, J. Am. Ceram. Soc. 82, 3299 (1999).
M. Henry, J.P. Jolivet, and J. Livage, in Chemistry, Spectroscopy and Applications of Sol-Gel Glasses, edited by R. Reisfeld and C.K. Jørgensen (Springer Verlag, Berlin, 1992), p. 153.
Handbook of Chemistry and Physics (The Chemical Rubber Co., Cleveland, OH, 1966).
R.K. Iler, The Chemistry of Silica (Wiley, New York, 1979).
I. Jaymes and A. Douy, J. Am. Ceram. Soc. 75, 3154 (1992).
A. Douy, P. Canale, and J. Coutures, J. Europ. Ceram. Soc. 9, 373 (1992).
A. Douy, J. Non-Cryst. Solids 147/148, 554 (1992).
I. Jaymes, A. Douy, M. Gervais, and J.P. Coutures, J. Sol-Gel Sci. & Technol. 8, 415 (1997).
I. Jaymes and A. Douy, J. Sol-Gel Sci. & Technol. 4, 7 (1995).
I. Jaymes, A. Douy, D. Massiot, and J.P. Busnel, J. Am. Ceram. Soc. 78, 2648 (1995).
C. Veytizou, J.-F. Quinson, and A. Douy, J. Mat. Chem. 10, 365 (2000).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Douy, A. Aqueous Syntheses of Forsterite (Mg2SiO4) and Enstatite (MgSiO3). Journal of Sol-Gel Science and Technology 24, 221–228 (2002). https://doi.org/10.1023/A:1015332607551
Issue Date:
DOI: https://doi.org/10.1023/A:1015332607551