Abstract
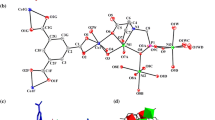
The title compounds were obtained from the reactions of copper or silver monohalides with the bidentate bis(diphenylphosphino)methane (dppm) ligand at room temperature in a mixed solvent. Single-crystal X-ray diffraction analyses indicate that both compounds crystallize in a monoclinic system but in different space groups. The structures are characterized by a trinuclear [M3I2(dppm)3]+ cation with a trigonal-bipyramid [M3(μ 3-I)2] core. In agreement with the geometric characteristics of the M3 triangles, 31P NMR spectra exhibit a single peak for the [Cu3] cluster but a double-peak for the [Ag3] cluster. Preliminary optical studies by UV/Vis and emission techniques show major absorption shoulders at ∼286 nm for Cu3I2(dppm)3·I and ∼254 nm for Ag3I2(dppm)3·I, but no luminescence in the non-degassed MeCN solution at 298 K. The structures, bonding, electronic excitations and emissions are discussed based on relativistic density functional theory calculations.
Similar content being viewed by others
References
P. C. Ford (1994).Coord. Chem. Rev. 132, 129.
P. C. Ford, E. Cariati, and J. Bourassa (1999). Chem. Rev. 99, 220.
V. W.-W. Yam and K. K. W. Lo (1999). Chem. Soc. Rev. 28,323.
M. R. Churchill and K. L. Kalra (1975). Inorg. Chem. 14,2502.
B.-K. Teo and J. C. Calabrese (1976). Inorg. Chem. 15, 2474.
D. D. Hardt and A. Pierre (1977). Inorg. Chim. Acta 25, L59; P. C. Ford and A. Vogler (1993). Acc. Chem. Res. 26, 220.
M. Henary and J. I. Zink (1991).Inorg. Chem. 30, 3111.
N. Bresciani, N. Marsich, G. Nardin, and L. Randaccio (1974). Inorg. Chim. Acta 10, L5.
J. K. Bera, M. Nethaji, and A. G. Samuelson (1999). Inorg. Chem. 38, 218.
J.-P. Lang and K. Tatsumi (1998). Inorg. Chem. 37, 6308; R.-N. Yang, Y.-H. Sun, X.-Y. Hu, and D.-M. Jin (1999). Chin. J. Chem. 17, 284.
V. J. Schubert, D. Neugebawer, and A. A. M. Aly (1980). Z. Anorg. Allg. Chem. 464, 217. A. A. M. Aly, D. Neugebauer, O. Orama, V. Schubert, and H. Schmidbaur (1978). Angew. Chem. Int. Ed. Engl. 17, 125.
D. Franzoni, G. Pelizzi, G. Predieri, P. Tarasconi, F. Vitali, and C. Pelizzi (1989). J. Chem. Soc., Dalton Trans. 247.
SADABS, Bruker Smart and Bruker Shelxtl Package, Bruker AXS GmbH, 1998.
G. M. Sheldrick, SHELXS-97, Program for X-Ray Crystal Structure Solution (Göttingen University, Germany, 1997).
G. M. Sheldrick, SHELXl-97, Program for X-Ray Crystal Structure Refinement (Göttingen University, Germany, 1997).
ADF 2000.02,Theoretical Chemistry, Vrije Universiteit, Amsterdam (http://www.scm. com). (a) E. J. Baerends, D. E. Ellis, and P. Ros (1973). Chem. Phys. 2, 41. (b)L. Versluis and T. Ziegler (1988). J. Chem. Phys. 88, 322. (c) G. te Velde and E. J. Baerends (1992). J. Comput. Phys. 99, 84. (d) C. Fonseca Guerra, J. G. Snijders, G. te Velde, and E. J. Baerends (1998). Theor. Chem. Acc. 99, 391.
(a)J. C. Slater, Quantum Theory of Molecular and Solids. Vol. 4: The Self-Consistent Field for Molecular and Solids (McGraw-Hill, New York, 1974). (b)S. H. Vosko, L. Wilk, M. Nusair (1980). Can. J. Phys. 58, 1200.
(a)J. P. Perdew and Y. Wang (1992). Phys Rev. B 45, 13244. (b)J. P. Perdew, J. A. Chevary, S. H. Vosko, K. A. Jackson, M. R. Pederson, D. J. Singh, and C. Foilhais (1992). Phys. Rev. B 46, 6671.
J. Li and K.-C. Wu (2000).Inorg. Chem. 39, 1538.
(a)J. G. Snijder, E. J. Baerends, and P. Vernooijs (1982). At. Nucl. Data Tables 26, 483. (b)P. Vernooijs, J. G. Snijder, and E. J. Baerends (1984). Slater Type Basis Functions for the Whole Periodic System, Internal Report, Free University of Amsterdam, The Netherlands.
T. Ziegler, E. J. Baerends, J. G. Snijder, and W. Ravenek (1989). J. Phys. Chem. 93, 3050.
S. J.A. van Gisbergen, J. G. Snijders, and E. J. Baerends (1999). Comp. Phys. Comm. 118, 119.
T. Ziegler, A. Rauk, and E. J. Baerends (1977). Theoret. Chim. Acta 43, 261.
V. W.-W. Yam and S. W. K. Choi (1996). J. Chem. Soc., Dalton Trans. 4227; D. Li, X. Hong, C. M. Che, W. C. Lo, and S. M. Peng (1993). J. Chem. Soc., Dalton Trans. 2929.
J. C. Vickery, M. M. Olmstead, E. Y. Fung, and A. L. Balch (1997). Angew. Chem. Int. Ed. Engl. 36, 1179.
J. Li and P. Pyykkö (1992). Chem. Phys. Lett. 197, 586.
V. W.-W. Yam, W.-K. Lee, K.-K. Cheung, B. Crystall, and D. Phillips (1996).J. Chem. Soc., Dalton Trans. 3283.
R. D. M. Ho and R. Bau (1983). Inorg. Chem. 22, 4079.
F. A. Cotton, G. Wilkinson, and P. L. Gauss, Basic Inorganic Chemistry, 3rd ed. (,Wiley, Toronto, 1995), p. 61.
R. Provencher and P. D. Harvey (1996). Inorg. Chem. 35, 2235.
V. W.-W. Yam, W. K. M. Fung, and K.-K. Cheung (1997). Organometallics 16, 2032.
N. Marsich, A. Camus, and E. Cebulec (1972). J. Inorg. Nucl. Chem. 34, 933.
G. B. Deacon and H. S. Green (1968). Spectrochimca Acta 24A, 845.
S. O. Grim and E. D. Walton (1980). Inorg. Chem. 19, 1982.
S. W. Carr and R. Colton (1981). Aust. J. Chem. 34, 35.
V. W.-W. Yam, W. K. M. Fung, and K. K. Cheung (1999). J. Cluster Sci. 10, 37.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhou, WB., Dong, ZC., Song, JL. et al. Syntheses, Structures, Bonding, and Optical Properties of Trinuclear Cluster Iodides: M3(μ3-I)2(μ-dppm)3·I (M=Cu, Ag), dppm=bis(diphenylphosphino)methane). Journal of Cluster Science 13, 119–136 (2002). https://doi.org/10.1023/A:1015195130685
Issue Date:
DOI: https://doi.org/10.1023/A:1015195130685