Abstract
Purpose. To gain insight into the molecular structure of amorphous compounds by investigating hydrogen bonding patterns and strength in a series of structurally related compounds. Seven 1,4-dihydro- pyridine calcium channel blockers were evaluated.
Methods. FT-Raman and FT-infrared spectra of the compounds in the crystalline and amorphous states were obtained.
Results. For crystalline compounds, the position of the NH vibration varied considerably, indicating that the strength of hydrogen bonding differs between the different compounds in agreement with published single crystal X-ray data. For the amorphous phases, the NH vibration occurred at approximately the same position for all compounds, suggesting a uniform average hydrogen bonding strength. Somewhat surprisingly, for some compounds, the average hydrogen bond strength in the amorphous state was found to be greater than in the crystalline compound, although for others, it was weaker as anticipated. Hydrogen bonding patterns (acceptor group) varied between the crystalline compounds, but were remarkably consistent in the amorphous compounds; thus the acceptor group in the amorphous phase is not necessarily the same as in the crystalline counterpart.
Conclusions. Hydrogen bond patterns and strength within a group of chemically related amorphous compounds were found to be very similar, but were different from those in the equivalent group of crystalline substances.
Similar content being viewed by others
REFERENCES
C. P. Brock and J. D. Dunitz. Towards a grammar of crystal packing. Chem. Mater. 6:1118–1127 (1994).
M. C. Etter. Encoding and decoding hydrogen-bond patterns of organic compounds. Acc. Chem. Res. 23:120–126 (1990).
R. H. Tromp, R. Parker, and S. G. Ring. A neutron scattering study of the structure of amorphous glucose. J. Chem. Phys. 107:6038–6049 (1997).
W. M. J. Risen. Optical spectra of glasses. J. Non-Cryst. Solids 76:97–108 (1985).
J. F. Carpenter, M. J. Pikal, B. S. Chang, and T. W. Randolph. Rational design of stable lyophilized protein formulations: Some practical advice. Pharm. Res. 14:969–975 (1997).
L. S. Taylor and G. Zografi. Spectroscopic characterization of interactions between pvp and indomethacin in amorphous molecular dispersions. Pharm. Res. 14:1691–1698 (1997).
L. S. Taylor and G. Zografi. Sugar-polymer hydrogen bond interactions in lyophilized amorphous mixtures. J. Pharm. Sci. 87:1615–1621 (1998).
P. C. Painter, Y. Park, and M. M. Coleman. Thermodynamics of hydrogen bonding in polymer blends. 1. The application of association models. Macromolecules 22:570–579 (1989).
C. Doherty and P. York. Evidence for solid-and liquid-state interactions in a furosemide-polyvinylpyrrolidone solid dispersion. J. Pharm. Sci. 76:731–737 (1987).
C. N. R. Rao and A. S. N. Murthy. Spectroscopic studies of hydrogen bonding. Develop. Appl. Spectrosc. 7B:54–88 (1970).
D. Lin-Vien, N. B. Colthup, W. G. Fateley, and J. G. Grasselli. The Handbook of Infrared and Raman Characteristic Frequencies of Organic molecules. Academic Press, Inc., San Diego, California, 1991.
K. Nakamoto, M. Margoshes, and R. E. Rundle. Stretching frequencies as a function of distances in hydrogen bonds. J. Amer. Chem. Soc. 77:6480–6486 (1955).
D. J. Skrovanek, S. E. Howe, P. C. Painter, and M. M. Coleman. Hydrogen bonding in polymers: Infrared temperature studies of an amorphous polyamide. Macromolecules 18:1676–1683 (1985).
G. A. Jeffrey. An Introduction to Hydrogen Bonding. Oxford University Press, New York, 1997.
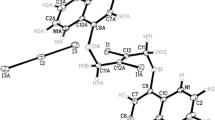
A. M. Triggle, E. Shefter, and D. J. Triggle. Crystal structures of calcium channel antagonists: 2,5-dimethyl-3,5-dicarbomethoxy-4[2-nitro-,3-cyano-,4-(dimethylamino)-, and 2,3,4,5,6-pentafluorophenyl]-1,4-dihydropyridine. J. Med. Chem. 23:1442–1445 (1980).
R. Fossheim. Crystal structure of the dihydropyridine ca2+ antagonist felodipine. Dihydropyridine binding prerequisites assessed from crystallographic data. J. Med. Chem. 29:305–307 (1986).
S. D. Wang, L. G. Herbette, and D. G. Rhodes. Structure of the calcium channel antagonist, nimodipine. Acta Cryst. C45:1748–1751 (1989).
R. Fossheim, A. Joslyn, A. J. Solo, E. Luchowski, A. Rutledge, and D. J. Triggle. Crystal structures and pharmacological activities of 1,4-dihydropyridine calcium channel antagonists of the isobutyl methyl 2,6-dimethyl-4-(substituted phenyl)-1,4-dihydropyridine-3,5-dicarboxylate (nisoldipine) series. J. Med. Chem. 31:300–305 (1988).
A. Burger, J. M. Rollinger, and P. Bruggeller. Binary system of (R)-and (S)-nitrendipine-polymorphism and structure. J. Pharm. Sci. 86:674–679 (1997).
R. C. Lord and R. E. Merrifield. Strong hydrogen bonds in crystals. J. Chem. Phys. 21:166–167 (1953).
P. Murray-Rust and J. P. Glusker. Directional hydrogen bonding to sp2 and sp3 hybridized oxygen atoms and its relevance to ligand macromolecular interactions. J. Am. Chem. Soc. 105:5761–5766 (1983).
R. Taylor, O. Kennard, and W. Versichel. Geometry of the NHO·C hydrogen bond. 3. Hydrogen bond distances and angles. Acta Cryst. B40:280–288 (1984).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Tang, X.C., Pikal, M.J. & Taylor, L.S. A Spectroscopic Investigation of Hydrogen Bond Patterns in Crystalline and Amorphous Phases in Dihydropyridine Calcium Channel Blockers. Pharm Res 19, 477–483 (2002). https://doi.org/10.1023/A:1015147729564
Issue Date:
DOI: https://doi.org/10.1023/A:1015147729564