Abstract
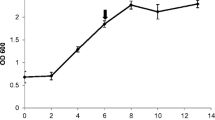
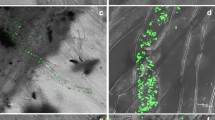
Defense responses of grapevine towards Botrytis cinerea were investigated. The expression of genes coding for proteins involved in defense were studied: (a) phenylalanine ammonia-lyase (PAL) and stilbene synthase (StSy), (b) an acidic chitinase (VCH3) and a basic chitinase (VCHIT1b), and (c) a polygalacturonase inhibitor protein (PGIP). Since no PGIP was known in grapevine, a complete cDNA sequence was first characterized by PCR and RACE-PCR amplifications. RNAs isolated from infected leaves and infected berries were analysed by semi-quantitative and real-time RT-PCRs. In infected leaves, the expression of PAL, StSy, PGIP and VCH3 genes occurred 6 hours post inoculation (hpi). Increase of VCHIT1b gene expression was delayed (24 hpi). Maximum levels of induction of these genes were observed at 48 hpi, except for the VCH3 gene (24 hpi). Activation of these defense responses was not sufficient to stop B. cinerea spread. In berries, no VCH3 gene expression was detected. Maximum levels of induction were observed in stage 3 (loss of berry colour and abundant production of conidia) for the PAL and PGIP genes, and in stage 4 (shrivelled berry) for the StSy and VCHIT1b genes.
Similar content being viewed by others
References
Altschul SF, Madden TL, Schaffer AA, Zhang JH, Zhang Z, Miller W and Lipman DJ (1997) Gapped BLAST and PSIBLAST: A new generation of protein database search programs. Nucleic Acids Research 25: 3389–3402
An YQ, McDowell JM, Huang S, McKinney EC, Chambliss S and Meagher RB (1996) Strong, constitutive expression of the Arabidopsis ACT2/ACT8 actin subclass in vegetative tissues. The Plant Journal 10: 107–121
Benito EP, ten Have A, van't Klosster JW and van Kan JAL (1998) Fungal and plant gene expression during synchronized infection of tomato leaves by Botrytis cinerea. European Journal of Plant Pathology 104: 207–220
Busam G, Kassemeyer HH and Matern U (1997a) Differential expression of chitinases in Vitis vinifera L. responding to systemic acquired resistance activators or fungal challenge. Plant Physiology 115: 1029–1038
Busam G, Junghanns KT, Kneusel RE, Kassemeyer HH and Matern U (1997b) Characterization and expression of caffeoylcoenzyme A 3-O-methyltransferase proposed for the induced resistance response of Vitis vinifera L. Plant Physiology 115: 1039–1048
Cervone F, Hahn M, De Lorenzo G, Darvill A and Albersheim P (1989) Host—Pathogen interactions XXXIII. A plant protein converts a fungal pathogenesis factor into an elicitor of plant defense responses. Plant Physiology 90: 542–548
Collinge DB and Slusarenko AJ (1987) Plant gene expression in response to pathogens. Plant Molecular Biology 9: 389–410
Davies C and Robinson SP (1996) Sugar accumulation in grape berries. Cloning of two putative vacuolar invertase cDNAs and their expression in grapevine tissues. Plant Physiology 111: 275–283
Davies C and Robinson SP (2000) Differential screening indicates a dramatic change in mRNA profiles during grape berry ripening. Cloning and characterization of cDNAs encoding putative cell wall and stress response proteins. Plant Physiology 122: 803–812
De Waard MA, Georgopoulos SG, Hollomon DW, Ishii H, Leroux P, Ragsdale NN and Schwinn FJ (1993) Chemical control of plant diseases: Problems and prospects. Annual Review of Phytopathology 31: 403–421
Derckel JP, Legendre L, Audran JC, Haye B and Lambert B (1996) Chitinases of the grapevine (Vitis vinifera L.): Five isoforms induced in leaves by salicylic acid are constitutively expressed in other tissues. Plant Science 119: 31–37
Derckel JP, Baillieul F, Manteau S, Audran JC, Haye B, Lambert B and Legendre L (1999) Differential induction of grapevine defenses by two strains of Botrytis cinerea. Phytopathology 89: 197–203
Elad Y(1997) Responses of plants to infection by Botrytis cinerea and novel means involved in reducing their susceptibility to infection. Biological Reviews 72: 381–422
Elad Y and Evensen K (1995) Physiological aspects of resistance to Botrytis cinerea. Phytopathology 85: 637–643
Gullino M (1992) Chemical control of Botrytis spp. In: Verhoeff K, Malathrakis NE and Williamson B (eds) Recent Advances in Botrytis Research (pp 217–222) Pudoc Scientific Publishers, Wageningen
Jacobs AK, Dry IB and Robinson SP (1999) Induction of different pathogenesis-related cDNAs in grapevine infected with powdery mildew and treated with ethephon. Plant Pathology 48: 325–336
Jarvis WR(1980) Epidemiology. In: Coley-Smith JR, Verhoeff K and Jarvis WR (eds) The Biology of Botrytis (pp 181–217) Academic Press, London
Jeandet P, Adrian M, Joubert JM, Hubert F and Bessis R (1996) Stimuler les défenses naturelles de la vigne. Un complément à la lutte phytosanitaire contre le Botrytis. Phytoma 488: 21–25
Jeandet P, Bessis R, Sbaghi M and Meunier P (1995) Production of the phytoalexin resveratrol by grapes as a response to Botrytis attacks in the vineyard. Journal of Phytopathology 143: 135–139
Kessmann H, Staub T, Hofmann C, Maetzke T, Herzog J, Ward E, Uknes S and Ryals J (1994) Induction of systemic acquired disease resistance in plants by chemicals. Annual Review of Phytopathology 32: 439–459
Kombrink E and Somssich IE (1995) Defense responses of plants to pathogens. Advances in Botanical Research 21: 1–34
Kraeva E, Tesnière C, Terrier N, Romieu C, Sauvage F, Bierne J and Deloire A (1998) Transcription of a ?-1,3-glucanase gene in grape berries in a late developmental period, or earlier after wounding treatment. Vitis 37: 107–111
Langcake P and McCarthy WV (1979) The relationship of resveratrol production to infection of grapevine leaves by Botrytis cinerea. Vitis 18: 244–253
Mazeyrat F, Mouzeyar S, Courbou I, Badaoui S, Roeckel-Drevet P, Tourvieille de Labrouhe D and Ledoigt G (1999) Accumulation of defense related transcripts in sun-flower hypocotyls (Helianthus annuus L.) infected with Plasmopara halstedii. European Journal of Plant Pathology 105: 333–340
Moniz de Sa M and Drouin G (1996) Phylogeny and substitution rates of angiosperm actin genes. Molecular Biology and Evolution 13: 1198–1212
Powell AL, van Kan J, ten Have A, Visser J, Greve LC, Bennett AB and Labavitch JM (2000) Transgenic expression of pear PGIP in tomato limits fungal colonization. Molecular Plant–Microbe Interactions 13: 942–950
Renault AS, Deloire A and Bierne J (1996) Pathogenesis-related proteins in grapevines induced by salicylic acid and Botrytis cinerea. Vitis 35: 49–52
Reuveni M (1998) Relationship between leaf age, peroxidase and ?-1,3-glucanase activity, and resistance to downy mildew in grapevines. Journal of Phytopathology 146: 525–530
Robinson SP, Jacobs AK and Dry IB (1997) A class IV chitinase is highly expressed in grape berries during ripening. Plant Physiology 114: 771–778
Salzman RA, Tikhonova I, Bordelon BP, Hasegawa PM and Bressan RA (1998) Coordinate accumulation of antifungal proteins and hexoses constitutes a developmentally controlled defense response during fruit ripening in grape. Plant Physiology 117: 465–472
Simpson CG, MacRae E and Gardner RC (1995) Cloning of a polygalacturonase-inhibiting protein from kiwifruit (Actinidia deliciosa). PGR95-037. Plant Physiology 108: 1748
Sparvoli F, Martin C, Scienza A, Gavazzi G and Tonelli C (1994) Cloning and molecular analysis of structural genes involved in flavonoid and stilbene biosynthesis in grape (Vitis vinifera L.). Plant Molecular Biology 24: 743–755
Steenkamp J, Wiid I, Lourens A and van Helden P (1994) Improved method for DNA extraction from Vitis vinifera. American Journal of Enology and Viticulture 45: 102–106
Stotz HU, Powell ALT, Damon SE, Greve LC, Bennett AB and Labavitch JM (1993) Molecular characterization of a polygalacturonase inhibitor from Pyrus communis L. cv Barlett. Plant Physiology 102: 133–138
Stotz HU, Contos JJA, Powell ALT, Bennett AB and Labavitch JM (1994) Structure and expression of an inhibitor of fungal polygalacturonases from tomato. Plant Molecular Biology 25: 607–617
Tattersall DB, van Heeswijck R and Hoj PB (1997) Identification and characterization of a fruit-specific, thaumatin-like protein that accumulates at very high levels in conjunction with the onset of sugar accumulation and berry softening in grapes. Plant Physiology 114: 759–769
ten Have A, Mulder W, Visser J and van Kan JAL (1998) The endopolygalacturonase gene Bcpg1 is required for full virulence of Botrytis cinerea. Molecular Plant–Microbe Interactions 11: 1009–1016
Toubart P, Desiderio A, Salvi G, Cervone F, Daroda L, de Lorenzo G, Bergmann C, Darvill AG and Albersheim P (1992) Cloning and characterization of the gene encoding the endopolygalacturonase-inhibiting protein (PGIP) of Phaseolus vulgaris L. The Plant Journal 2: 367–373
Winer J, Jung CKS, Shackel I and Williams PM (1999) Development and validation of real-time quantitative reverse transcriptase-polymerase chain reaction for monitoring gene expression in cardiac myocytes in vitro. Analytical Biochemistry 270: 41–49
Yao C, Conway WS, Ren R, Smith D, Ross GS and Sams CE (1999) Gene encoding polygalacturonase inhibitor in apple fruit is developmentally regulated and activated by wounding and fungal infection. Plant Molecular Biology 39: 1231–1241
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bézier, A., Lambert, B. & Baillieul, F. Study of Defense-related Gene Expression in Grapevine Leaves and Berries Infected with Botrytis cinerea . European Journal of Plant Pathology 108, 111–120 (2002). https://doi.org/10.1023/A:1015061108045
Issue Date:
DOI: https://doi.org/10.1023/A:1015061108045