Abstract
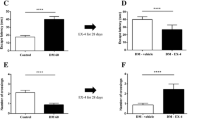
The endothelial or epithelial tight junctions create a barrier to diffusion of solutes. Since experimental diabetes mellitus is associated with considerable alterations in the blood-brain barrier (BBB), it is possible that specific tight junction proteins may be altered in diabetes. To test this hypothesis, Western and Northern blot analysis were carried out to measure the steady-state level of occludin and zonula occludens-one (ZO-1) proteins and mRNA levels in cerebral tissue of streptozotocin-induced diabetic rats and the results were compared to insulin treated diabetic rats and vehicle injected control rats. The cerebral occludin content in diabetic rats (115.4 ± 18.6 arbitrary units) was significantly reduced compared to insulin-treated diabetic rats (649.1 ± 141.2) or control rats (552.9 ± 82.9), p < 0.001. The ZO-1 content of cerebral tissue from diabetic rats (1240.6 ± 199.7 arbitrary units) was not significantly altered compared to controls (1310.8 ± 256.9). The cerebral occludin mRNA content relative to G3PDH mRNA was 1.35 ± 0.07 and 1.34 ± 0.19 in control and diabetic rats respectively. The cerebral ZO-1 mRNA content relative to G3PDH mRNA in diabetic and control rats was 1.135 ± 0.123 and 0.956 ± 0.038 respectively. These differences did not achieve statistical significance. It is concluded that diabetes alters the molecular anatomy of the tight junctions in cerebral tissue by altering the content of select structural proteins.
Similar content being viewed by others
REFERENCES
Mitic, L. L. and Anderson, J. M. 1998. Molecular architecture of tight junctions. Annu. Rev. Physiol. 60:121–142.
Hirase, T., Staddon, J. M., Saitou, M., Ando-Akatsuka, Y., Itoh, M., Furuse, M., Fujimoto, K., Tsukita, S., and Rubin, L. L. 1997. Occludin as a possible determanant of tight junction permeability in endothelial cells. J. Cell. Sci. 110:1603–1613.
Fruse, M., Hirase, T., Itoh, M., Nagafuchi, A., Yonemura, S., Tsukita, S., and Tsukita, S. 1993. Occludin: a novel integral membrane protein localizing at tight junctions. J. Cell Biol. 123:1631–1633.
Mooradian, A. D. 1988. Diabetic complications of the central nervous system. Endocr. Rev. 9:346–356.
Mooradian, A. D. 1997. Pathophysiology of central nervous system disorders in diabetes. Clin. Neurosci. 4:322–326.
Mooradian, A. D. 1997. Central nervous system complications of diabetes. A perspective from the blood-brain barrier. Brain Res. Rev. 23:210–218.
Mooradian, A. D. and Scarpace, P. J. 1992. Beta adrenergic receptor activity of cerebral microvessels in experimental diabetes mellitus. Brain Res. 583:155–160.
Mooradian, A. D., Grabau, G., and Uko-eninn, A. 1993. In situ quantitative estimates of the age-related and diabetes-related changes in cerebral endothelial barrier antigen. Brain Res. 309:41–44.
Mooradian, A. D. 1992. Altered chromatin structure of cerebral nuclei in experimental diabetes mellitus. Proc. Soc. Exp. Biol. Med. 199:282–286.
Ando-Akatsuka, Y., Saitou, M., Hirase, T., et al. 1996. Interspecies diversity of the occludin sequence: cDNA cloning of human, mouse, dog, and rat-kangaroo homologues. J. Cell Biol. 133:43–47.
Lowry, O. H., Rosebrough, N. J., Farr, A. L., and Randall, R. J. 1951. Protein measurement with the folin phenol reagent. J. Biol. Chem. 193:265–275.
Laemmli, U. K. 1970. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227:680–685.
Towbin, H., Staehelin, T., and Gordon, J. 1979. Electrophoretic transfer of proteins from polyacrylamide gels to nitrocellulose sheets. Proc. Nat'l. Acad. Sci. USA 76:4350–4354.
Chirgwin, J. M., Przybylz, A. E., MacDonald, R. J., and Rutter, W. J. 1979. Isolation of biologically active ribonucleic acid from sources enriched in ribonuclease. Biochemistry 18:5294–5299.
Sambrook, J., Frisch, E. F., and Maniatis. T. 1989. Molecular Cloning: A Laboratory Manual. Ed. 2, Cold Spring Harbor Laboratory, Cold Spring Harbor, New York.
Balda, M. S. and Anderson, J. M. 1993. Two classes of tight junctions are revealed by ZO-1 isoforms. Am. J. Physiol. 264: C918–C924.
Knissel, U. and Hartwig, W. 2000. Tight junctions of the bloodbrain barrier. Cell. Molecule. Neurobiol. 20:57–76.
Antonetti, D. A., Barker, A. J., Khin, S. et al. 1998. Vascular permeability in experimental diabetes is associated with reduced endothelial occludin content. Vascular endothelial growth factor decreases occludin in retinal endothelial cells. Diabetes 47:1953–1959.
Gardner, T. W. 1995. Histamine, ZO-1 and increased bloodretinal barrier permeability in diabetic retinopathy. Trans. Am. Ophtalmol. Soc. 93:583–621.
Maiello, M., Boeri, D., Podesta, F., et al. 1992. Increased expression of tissue plasminogen activator and its inhibitor and reduced fibrinolytic potential of human endothelial cells cultured in elevated glucose. Diabetes 41:1009–1015.
Cagliero, E., Roth, T., Rot, S., Maiello, M., and Lorenzi, M. 1991. Expression of genes related to extracellular matrix in human endothelial cells. Differential modulation by elevated glucose concentrations, phorbol esters, and cAMP. J. Biol. Chem. 266:14244–14250.
Lorenzi, M., Montisano, D. F., Toledo, S., and Barrieux, A. 1986. High glucose induces DNA damage in cultured human endothelial cells. J. Clin. Invest. 77:322–325.
Mooradian, A. D., Pinnas, J. L., Lung, C. C., Yahya, D., and Meredith, K. 1994. Diabetes-related changes in the protein composition of rat cerebral microvessels. Neurochem. Res. 19: 123–128.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Chehade, J.M., Haas, M.J. & Mooradian, A.D. Diabetes-Related Changes in Rat Cerebral Occludin and Zonula Occludens-1 (ZO-1) Expression. Neurochem Res 27, 249–252 (2002). https://doi.org/10.1023/A:1014892706696
Issue Date:
DOI: https://doi.org/10.1023/A:1014892706696