Abstract
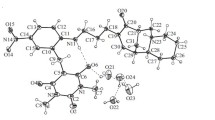
The reaction of barbituric acid and its N-substituted derivatives and 2-thio analogs with cotarnine forms 5-(4-methoxy-6-methyl-5,6,7,8-tetrahydro-2H-1,3-methylenedioxy-[4,5-g]isoquinolinyl-1)barbituric acids, a new class of zwitter-ions, the structure of which was studied by 1 H and 13 C NMR spectroscopy and mass spectrometry. The prepared compounds exist in solution as stable intermolecular associates and have a complicated H-bonded structure.
Similar content being viewed by others
REFERENCES
A. P. Orekhov, Alkaloid Chemistry [in Russian], ONTI, Moscow (1938), p. 622.
D. Beke, in: Advances in Heterocyclic Chemistry, (1963), A. R. Katritzky, ed., Academic Press, New York (1963), Vol. 1, p. 167.
H. Mohrle and B. Grimm, Arch. Pharm. (Weinheim), 319, 835 (1986).
M. Chrzanovska and A. Sulma, Pol. J. Chem., 72, 2202 (1998).
K. W. Bentley, Nat. Prod. Rep., 9, 555 (1992).
K. A. Krasnov, M. N. Yurova, and V. G. Kartsev, in: Nitrogen-Containing Heterocycles and Alkaloids, V. G. Kartsev and G. A. Tolstikov, eds., Iridium Press, Moscow (2001), Vol. 2, p. 152.
B. A. Ivin, A. D. D'yachkov, I. M. Vishnyakov, N. A. Smorygo, and E. G. Sochilin, Zh. Org. Khim., 11, 1337 (1979).
E. Haslinger, M. Reithmaier, W. Robien, and P. Wolschann, Monatsh. Chem., 115, 375 (1984).
A. I. Biggs, J. Chem. Soc., 2485 (1956).
K. A. Krasnov and V. I. Slesarev, Zh. Org. Khim., 4, 601 (1997).
H. G. Mantner and E. M. Clauton, J. Am. Chem. Soc., 23, 6270 (1959).
E. Fisher and A. Diltey, Liebigs Ann. Chem., 335, 334 (1904).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Krasnov, K.A., Kartsev, V.G. & Yurova, M.N. Chemical Modification of Plant Alkaloids. 2. Reaction of Cotarnine with Barbituric Acid Derivatives and Structure of 5-Dihydrocotarnylbarbituric Acids. Chemistry of Natural Compounds 37, 543–550 (2001). https://doi.org/10.1023/A:1014821016904
Issue Date:
DOI: https://doi.org/10.1023/A:1014821016904