Abstract
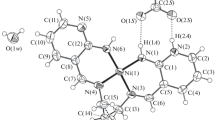
Metal complexes of general formula M(L)X2 and M(L)X3 [L = (4S,5S)-2,2-dimethyl-4,5-bis{6-[(4,5-dihydro-4-(S)-(1-methylethyl)oxazol-2-yl)pyridin-2-yl]}-1,3-dioxolane] were obtained by reacting, respectively, CoII, CuII, NiII, and ZnII nitrate salts and the RhIII chloride salt, with a chiral C2-symmetric bis(oxazolinylpyridinyl)dioxolane (L) ligand, in MeOH/CHCl3 solution. A single crystal X-ray analysis was carried out on [Ni(L)(OH2)2](NO3)2 · 2H2O and the molecular structure of L was also determined. In the free ligand the two symmetric arms are essentially planar and oriented nearly perpendicular to the dioxolane average plane. In the Ni complex one seven-membered and two five-membered chelation rings are formed. The metal atom also lies on the C2 axis, and two symmetry-related water molecules complete the octahedral coordination environment. Both compounds crystallize in chiral space groups; the ligand crystallizes in orthorhombic system, space group C 2 2 21, Z = 4; the nickel complex crystallizes in tetragonal system, space group P 43 21 2, Z = 4.
Similar content being viewed by others
References
T.G. Gant and A.I. Meyers, Tetrahedron, 50, 2297 (1994).
H. Nishiyama, S. Yamaguchi, S. Park and K. Itoh, Tetrahedron: Asymmetry, 1, 143 (1993).
J.S. Johnson and D.A. Evans, Acc. Chem. Res., 33, 325 (2000).
H. Brunner and R. Störiko, Europ. J. Inorg. Chem., 6, 783 (1998).
G. Chelucci, Tetrahedron: Asymmetry, 8, 2667 (1997).
A. Altomare, G. Cascarano, C. Giacovazzo, A. Guagliardi, M.C. Burla, G. Polidori and M. Camalli, J. Appl. Cryst., 27, 435 (1994).
G.M. Sheldrick. SHELXL97. Program for structure refinement., University of Goettingen, Germany, 1997.
D. Cremer and J.A. Pople, J. Am. Chem. Soc., 97, 1354 (1975).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Bacchi, A., Pelizzi, G., Minardi, G. et al. Coordinating properties of C2-symmetric chiral bis(oxazolinylpyridinyl)dioxolane ligands. Transition Metal Chemistry 27, 274–278 (2002). https://doi.org/10.1023/A:1014815000438
Issue Date:
DOI: https://doi.org/10.1023/A:1014815000438