Abstract
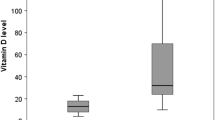
Several lines of evidence point to a possible relationship between vitamin D and cardiovascular disease. Animal experiments and observational studies in humans suggest vitamin D to be arteriotoxic and an association of high intake of vitamin D with increased incidence of ischemic heart disease (IHD). The major source of vitamin D in adults is vitamin D synthesized in the skin through exposure to the sun. In tropical environment there is a possibility of high level of solar exposure and enhanced serum levels of vitamin D in the population. We explored the relation between serum level of 25-hydroxyvitamin D3 and IHD in a case-control study involving 143 patients with either angiographic evidence of coronary artery disease or patients with acute myocardial infarction and 70 controls, all men in the age group of 45–65 years. Fasting blood samples were collected, serum separated and serum levels of 25-hydroxyvitamin D3 was measured by protein binding radioligand assay. Serum levels of cholesterol, triglyceride, calcium, magnesium and inorganic phosphate were also determined. Prevalences of diabetes, hypertension and smoking history were noted. Statistical comparisons of variables between cases and controls were done using χ2-tests. Multivariate logistic regression analysis was done to examine the association of IHD with serum levels of 25-hydroxyvitamin D3 controlling for selected variables. Serum levels of 25-hydroxyvitamin D3, calcium, inorganic phosphate, total cholesterol, low density lipoprotein and triglycerides were elevated in a higher proportion of patients, compared to controls. Serum levels of 25-OH-D3 above 222.5 nmol/l (89 ng/ml) was observed in 59.4% of cases compared to 22.1% in controls (p < 0.001; unadjusted odds ratio (OR): 5.17; 95% confidence interval (CI): 2.62–10.21). When controlled for age and selected variables using the multivariate logistic regression, the adjusted OR relating elevated serum 25-hydroxyvitamin D3 levels ( ≥222.5 nmol/l, ≥89 ng/ml) and IHD is 3.18 (95% CI: 1.31–7.73). Given the evidences for the arteriotoxicity of vitamin D, further investigations are warranted to probe whether the elevated serum levels of 25-hydroxyvitamin D3 observed in patients with IHD in a tropical environment has any pathogenic significance.
Similar content being viewed by others
References
Koh E, Morimoto S, Fukuo K, et al. 1,25-dihydroxyvitamin D3 binds speci.cally to rat vascular smooth muscle cells and stimulate their proliferation in vitro. Life Sciences 1988; 42: 215–223.
Seelig MS. Vitamin D and cardiovascular, renal and brain damage in infancy and childhood. Ann NY Acad Sci 1969; 147: 539–582.
Hughes MR, Baylink DJ, Jones PG, Haussler MR. Radioligand receptor assay for 25-hydroxyvitamin D2/ D3 and 1,25-dihydroxyvitamin D2/D3. J Clin Invest 1976; 58: 61–70.
Bajwa GS, Morrison LM, Ershoff BH. Induction of aortic and coronary athero-arteriosclerosis in rats fed a hypervitaminosis D, cholesterol-containing diet. Proc Soc Exp Biol Med 1971; 138: 975–982.
Toda T, Leszczynski DE, Kummerow FA. The role of 25-hydroxyvitamin D3 in the induction of atherosclerosis in swine and rabbit by hypervitaminosis D. Acta Pathol Jpn 1983; 33: 37–44.
Liu LB, Taylor CB, Peng SK, Mikkelson B. Experimental arteriosclerosis in Rhesus monkeys induced by multiple risk factors: Cholesterol, vitamin D and nicotine. Arterial Wall 1979; 5: 25–32.
Linden V. Vitamin D and myocardial infarction. Br Med J 1974; 3: 647–658.
Moon J, Bandy B, Davison AJ. Hypothesis: Etiology of atherosclerosis and osteoporosis: Are imbalances in the calciferol endocrine system implicated? J Am Coll Nutr 1992; 11: 567–583.
Davies H. Coronary heart disease. The signi.cance of coronary pathology in infancy and the role of mitogens such as vitamin D. Med Hypothesis. 1989; 30: 179–185.
National Nutrition Monitoring Bureau Report, National Institute of Nutrition, Hyderabad, India, 1994.
Raman Kutty V, Balakrishnan KG, Jayasree AK, Thomas J. Prevalence of coronary heart disease in the rural population of Thiruvananthapuram district, Kerala. Int J Cardiol 1993; 39: 59–70.
Rose GA, Blackburn H, Gillum RG, Prineas RJ. Cardiovascular survey methods. Geneva: World Health Organization, 1982.
National high blood pressure education program working group report on primary prevention of hypertension, US department of health and human services. 1993, NIH publication no. 93–2669.
National Cholesterol Education Program. Second report of the expert panel on detection, evaluation and treatment of high blood cholesterol in adults. Circulation 1994; 89: 1329–1445.
Chen TC, Adrian KT, Holick MF. Methods for the determination of the circulating concentrations of 25-hydroxyvitamin D. J Nutr Biochem 1990; 1: 315–319.
Gimblet EG, Marney AF, Bonsnes RW. Determination of calcium and magnesium in serum, urine, diet and stool by atomic absorption spectrophotometry. Clin Chem 1967; 13: 204–214.
Bostick RM, Kushi LH, Wu Y, Meyer KA, Sellers TA, Folsom R. Relation of calcium, vitamin D and dietary food intake to ischemic heart disease mortality among post menopausal women. Am J Epidemiol 1999; 149: 151–161.
Schmidt GH, Goossen J, Lindle F, Seidel D. Serum 25-hydroxy cholecalciferol in myocardial infarction. Atherosclerosis 1977; 26: 55–58.
Scragg R, Jackson R, Holdaway IM, Lim T, Beaglehole R. Myocardial Infarction is inversely associated with plasma 25-hydroxyvitamin D3 levels. A community based study. Int J Epidemiol 1990; 19: 59–63.
Scragg R, Holdaway I, Jackson R, Lim T. Plasma 25-hydroxyvitamin D3 and its relation to physical activity and other heart disease risk factors in the general population. Ann Epidemiol 1992; 2: 697–703.
Shepard RM, DeLuca HF. Plasma concentration of vitamin D3 and its metabolites in the rat as infuenced by vitamin D3 or 25-hydroxyvitamin D3 intakes. Arch Biochem Biophys 1980; 202: 43–53.
Holick MF. Vitamin D – Solar rays, the milky way, or both? New Engl J Med 1992; 326: 1213–1214.
Loomis WF. Skin pigment regulation of vitamin D biosynthesis in man. Science 1967; 57: 501–506.
Holick MF, Mac Laughlin JA, Doppeet SH. Regulation of cutaneous previtamin D3 photosynthesis in man: Skin pigment is not an essential regulator. Science 1981; 211: 590–592.
Sedrani SH, Elidrissy T, El Arabi KM. Sunlight and vitamin D status in normal Saudi subjects. Am J Clin Nutr 1983; 38: 129–132.
Matsuoka LY, Wortsman J, Hollis BW. Sun tanning and cutaneous synthesis of vitamin D3. J Lab Clin Med 1990; 116: 87–90.
Shepard R, Horst RL, Hamstra, De Luca. Determination of vitamin D and its metabolites in plasma from normal and anephric man. Biochem J 1979; 182: 55–69.
Haddad JG, Chyu KJ. Competitive protein binding radio assay for 25-hydroxycholecalciferol. J Clin Endocrinol 1971; 33: 992–995.
Stamp TCB. Factors in human vitamin D nutrition and in the production and cure of classical rickets. Proc Nutr Soc 1975; 34: 119–130.
Clemens TL, Henderson SL, Adams JS, Holick MF. Increased skin pigment reduces the capacity of skin to synthesise vitamin D3. Lancet 1982; 1: 74–76.
Stamp TCB, Haddad TG, Twigg CA. Comparison of oral 25-hydroxy-cholecalciferol, vitamin D and ultraviolet light as determinants of circulating 25-hydroxyvitamin D. Lancet 1977; 1: 1341–1343.
Kummerow FA. Nutrition, imbalance and angiotoxins as dietary risk factors in coronary heart disease. Am J Nutr 1979; 32: 58–83.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Rajasree, S., Rajpal, K., Kartha, C. et al. Serum 25-hydroxyvitamin D3 levels are elevated in South Indian patients with ischemic heart disease. Eur J Epidemiol 17, 567–571 (2001). https://doi.org/10.1023/A:1014559600042
Issue Date:
DOI: https://doi.org/10.1023/A:1014559600042