Abstract
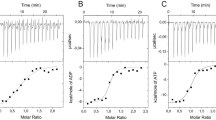
The kinetics of hydrolysis of the inorganic (PPi) and organic (ATP) substrates by Escherichia coli inorganic pyrophosphatase (PPase) and its mutant forms with Asp42 replaced by Ala, Asn, or Glu was studied. The Mn2+ or Zn2+ ions were used as activators of the enzymatic reaction. The kinetic parameters of hydrolysis were determined. The inhibitory effect of these cations on substrate hydrolysis was investigated. The dissociation constants were calculated for the Mn2+- and Zn2+-binding activator and inhibitor subsites of E. coli PPase. The observed hydrolysis rate of PPi increases in the series Zn2+ < Mn2+ < Mg2+, whereas the potential efficiency of these cations decreases in this series. Hydrolysis of ATP by E. coli PPase occurs only in the presence of Mn2+. The reasons for the observed differences in the substrate specificity of the enzyme are discussed.
Similar content being viewed by others
References
M. Kunitz, J. Gen. Physiol., 1952, 35, 423.
M. J. Schlesinger and M. J. Coon, Biochim. Biophys. Acta, 1960, 41, 30.
B. S. Cooperman, Methods Enzymol., 1982, 87, 526.
V. Yu. Oganessyan, S. A. Kurilova, N. N. Vorobyeva, T. I. Nazarova, A. N. Popov, A. A. Lebedev, S. M. Avaeva, and E. H. Harutyunyan, FEBS Lett., 1994, 348, 301.
J. Käpylä, T. Hyytiä, R. Lahti, A. Goldman, A. A. Baykov, and B. S. Cooperman, Biochemistry, 1995, 34, 792.
A. A. Baykov and A. S. Shestakov, Eur. J. Biochem., 1992, 206, 463.
E. H. Harutyunyan, I. P. Kuranova, B. K. Vainshtein, W. E. Höhne, V. S. Lamzin, L. Dauter, A. V. Teplyakov, and K. S. Wilson, Eur. J. Biochem., 1996, 239, 220.
P. Heikinheimo, J. Lehtonen, A. Baykov, R. Lahti, B. S. Cooperman, and A. Goldman, Structure, 1996, 4, 1491.
E. H. Harutyunyan, V. Yu. Oganessyan, N. N. Oganessyan, S. M. Avaeva, T. I. Nazarova, N. N. Vorobyeva, S. A. Kurilova, R. Huber, and T. Mather, Biochemistry, 1997, 36, 7754.
S. M. Avaeva, N. N. Vorob'eva, S. A. Kurilova, T. I. Nazarova, K. M. Polyakov, E. V. Rodina, and V. R. Samygina, Biokhimiya, 2000, 65, 442 [Biochemistry USSR, 2000, 65 (Engl. Transl.)].
A. V. Kuznetsov, S. M. Avaeva, A. A. Baykov, and V. A. Sklyankina, Biokhimiya, 1977, 42, 950 [Biochemistry USSR, 1977, 42 (Engl. Transl.)].
M. S. Mel'nik, T. I. Nazarova, and S. M. Avaeva, Biokhimiya, 1982, 47, 323 [Biochemistry USSR, 1982, 47 (Engl. Transl.)].
S. A. Kurilova, A. V. Bogdanova, T. I. Nazarova, and S. M. Avaeva, Bioorg. Khim. 1984, 10, 1147 [Sov. J. Bioorg. Chem., 1982, 10 (Engl. Transl.)].
A. S. Shestakov, Ph. D. (Chem.) Thesis, Department of Chemistry, M. V. Lomonosov Moscow State University, Moscow, 1991 (in Russian).
M. S. Mel'nik, T. I. Nazarova, and S. M. Avaeva, Bioorg. Khim. 1984, 10, 1483 [Sov. J. Bioorg. Chem., 1984, 10 (Engl. Transl.)].
S. M. Avaeva, E. V. Rodina, S. A. Kurilova, T. I. Nazarova, and N. N. Vorobyeva, FEBS Lett., 1996, 392, 91.
J. Josse, J. Biol. Chem., 1966, 241, 1938.
S. M. Avaeva, E. V. Rodina, S. A. Kurilova, T. I. Nazarova, N. N. Vorobyeva, E. H. Harutyunyan, and V. Yu. Oganessyan, FEBS Lett., 1995, 347, 44.
A. A. Baykov and S. M. Avaeva, Anal. Biochem., 1981, 116, 1.
R. Phillips, Chem. Rev., 1966, 66, 501.
G. S. Baldwin, R. B. Sessions, S. G. Erskine, and S. E. Halford, J. Mol. Biol., 1999, 288, 87.
B. S. Cooperman, A. Panackal, B. Springs, and D. Hamm, Biochemistry, 1981, 20, 6051.
S. E. Volk, A. A. Baykov, and S. M. Avaeva, Biokhimiya, 1981, 46, 30 [Biochemistry USSR, 1981, 46 (Engl. Transl.)].
K. M. Welsh, A. Jakobyansky, B. Springs, and B. S. Cooperman, Biochemistry, 1983, 22, 2243.
W. F. Bosron, R. A. Anderson, M. C. Falk, F. C. Kenhey, and B. L. Vallee, Biochemistry, 1977, 16, 610.
E. A. Braga, A. A. Baykov, and S. M. Avaeva, Biokhimiya, 1973, 38, 344 [Biochemistry USSR, 1973, 38 (Engl. Transl.)].
S. A. Kurilova, T. I. Nazarova, and S. M. Avaeva, Bioorg. Khim. 1984, 10, 1336 [Sov. J. Bioorg. Chem., 1984, 10 (Engl. Transl.)].
H. Sigel, R. Tribolet, R. Malini-Balakrishan, and R. B. Martin, Inorg. Chem., 1987, 26, 2149.
E. V. Rodina and Yu. P. Vainonen, Tez. dokl. Mezhdunarodnaya Shkola-Konferentsiya “Gorizonty fiziko-khimicheskoi biologii” [Abstrs. of Papers, International School Conference on Prospects of Physicochemical Biology], Pushchino, Moscow region, 2000, 2, 103 (in Russian).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Vainonen, Y.P., Rodina, E.V., Vorobyeva, N.N. et al. Some features of hydrolysis of organic and inorganic substrates by Escherichia coli inorganic pyrophosphatase in the presence of various activator cations. Russian Chemical Bulletin 50, 1877–1884 (2001). https://doi.org/10.1023/A:1014394415781
Issue Date:
DOI: https://doi.org/10.1023/A:1014394415781